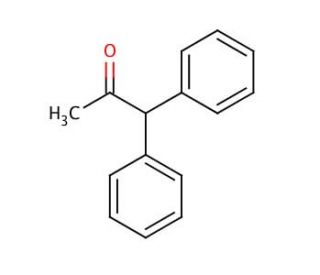
Molecular structure of Diphenylacetic acid, CAS Number: 117-34-0
Diphenylacetic acid (CAS 117-34-0)
CAS Number:
117-34-0
Molecular Weight:
212.24
Molecular Formula:
(C6H5)2CHCO2H
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Diphenylacetic acid is a compound that functions as a precursor in the synthesis of various organic compounds. At the molecular level, diphenylacetic acid undergoes specific chemical reactions to form new compounds with desired properties. Its mechanism of action involves participating in organic synthesis processes, where it serves as a starting material for the creation of more complex molecules. Diphenylacetic Acid plays a role in the development of diverse compounds through its involvement, contributing to the creation of new substances with potential applications in various fields.
Diphenylacetic acid (CAS 117-34-0) References
- [Inhibition of procaine hydrolysis by the diethylaminoethyl esters of diphenylpropylacetic acid (SKF 525-A) & of diphenylacetic acid (trasentin)]. | NETTER, KJ. 1959. Naunyn Schmiedebergs Arch Exp Pathol Pharmakol. 235: 498-512. PMID: 13657204
- SPreparation and pharmacological properties of some benzyl derivatives of diphenylacetic acid. | JOHNSEN, UE., et al. 1962. J Pharm Sci. 51: 799-801. PMID: 14036963
- Pd(II)-catalyzed enantioselective C-H olefination of diphenylacetic acids. | Shi, BF., et al. 2010. J Am Chem Soc. 132: 460-1. PMID: 20017549
- In vivo modulation of the inflammatory response by nonsteroidal antiinflammatory drug-related compounds that trigger L-selectin shedding. | Herrera-García, A., et al. 2013. Eur J Immunol. 43: 55-64. PMID: 22975861
- Kinetic resolution of racemic 2-hydroxy-γ-butyrolactones by asymmetric esterification using diphenylacetic acid with pivalic anhydride and a chiral acyl-transfer catalyst. | Nakata, K., et al. 2013. Org Lett. 15: 1170-3. PMID: 23461674
- Effect of renal dysfunction on the individual components of the acyl-glucuronide futile cycle. | Sallustio, BC., et al. 1989. J Pharmacol Exp Ther. 251: 288-94. PMID: 2795462
- Bis-Rhodamines Bridged with a Diazoketone Linker: Synthesis, Structure, and Photolysis. | Shojaei, H., et al. 2022. J Org Chem. 87: 56-65. PMID: 34919387
- [Methods of analyzing drug preparations of the group of diphenylacetic acid ethers]. | Adeĭshvili, LV. 1978. Farm Zh. 43-6. PMID: 365571
- Metabolism of arylacetic acids. 3. The metabolic fate of diphenylacetic acid and its variation with species and dose. | Dixon, PA., et al. 1977. Xenobiotica. 7: 717-25. PMID: 414462
- Synthesis and pharmacologic properties of the N-oxide of the diethylaminoethyl ester of diphenylacetic acid (N-oxide of adiphenine). | Patkowski, J. and Pomorski, J. 1966. Arch Immunol Ther Exp (Warsz). 14: 516-23. PMID: 4380616
- [Spectrophotometric studies of the esters of phenylhydroxyacetic acid, (I), diphenylhydroxyacetic acid (II) and diphenylacetic acid (3)]. | Zyzynski, W. 1971. Acta Pol Pharm. 28: 223. PMID: 5092585
- Determination of adiphenine, diphenylacetic acid and diethylamino-ethanol by high-performance liquid chromatography. | Michelot, J., et al. 1983. J Chromatogr. 257: 395-9. PMID: 6133881
- Enterohepatic recycling of phenolphthalein, morphine, lysergic acid diethylamide (LSD) and diphenylacetic acid in the rat. Hydrolysis of glucuronic acid conjugates in the gut lumen. | Parker, RJ., et al. 1980. Xenobiotica. 10: 689-703. PMID: 7445530
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Diphenylacetic acid, 5 g | sc-257376 | 5 g | $20.00 | |||
Diphenylacetic acid, 100 g | sc-257376A | 100 g | $91.00 |
