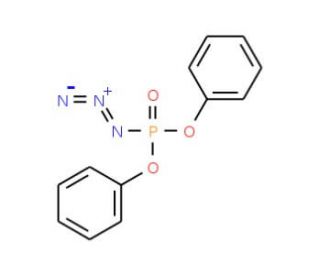
Molecular structure of Diphenyl phosphoryl azide, CAS Number: 26386-88-9
Diphenyl phosphoryl azide (CAS 26386-88-9)
Alternate Names:
DPPA; Phosphoric acid diphenyl ester azide
Application:
Diphenyl phosphoryl azide is a hydroazidation catalyst for facile preparation of organoazides
CAS Number:
26386-88-9
Molecular Weight:
275.20
Molecular Formula:
C12H10N3O3P
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Diphenyl phosphoryl azide is used in the aziridination of olefins catalyzed by colbalt-tetraphenylporphyrin. It is also used as the activating agent in the preparation of macrocyclic lactams and of an aldose reductase inhibitor. Used in the preparation of amino acid copolymers.
Diphenyl phosphoryl azide (CAS 26386-88-9) References
- Synthesis and characterization of novel polyurethane cationomers with dipeptide sequences and alkylammonium groups. | Buruiana, EC. and Buruiana, T. 2004. J Biomater Sci Polym Ed. 15: 781-95. PMID: 15255526
- Organization of collagen in the presence of diphenyl phosphoryl azide (DPPA): an in vitro study. | Usha, R., et al. 2013. Colloids Surf B Biointerfaces. 109: 121-8. PMID: 23624280
- Boc-protected 1-(3-oxocycloalkyl)ureas via a one-step Curtius rearrangement: mechanism and scope. | Sun, X., et al. 2014. Tetrahedron Lett. 55: 842-844. PMID: 24563555
- Synthesis of 1,2-dihydro-2-oxo-4-quinolinyl phosphates from 2-acyl-benzoic acids. | He, X., et al. 2015. Tetrahedron Lett. 56: 1441-1444. PMID: 25937677
- Liquid-Phase Vibrational Strong Coupling. | George, J., et al. 2015. J Phys Chem Lett. 6: 1027-31. PMID: 26262864
- Synthesis and inhibitory properties of some carbamates on carbonic anhydrase and acetylcholine esterase. | Yılmaz, S., et al. 2016. J Enzyme Inhib Med Chem. 31: 1484-91. PMID: 26985691
- Analogs of oxytocin containing a pseudopeptide Leu-Gly bond of cis and trans configuration. | Lebl, M., et al. 1989. Int J Pept Protein Res. 33: 16-21. PMID: 2722396
- Improved synthesis of glycine, taurine and sulfate conjugated bile acids as reference compounds and internal standards for ESI-MS/MS urinary profiling of inborn errors of bile acid synthesis. | Donazzolo, E., et al. 2017. Chem Phys Lipids. 204: 43-56. PMID: 28300538
- Nile tilapia skin collagen sponge modified with chemical cross-linkers as a biomedical hemostatic material. | Sun, L., et al. 2017. Colloids Surf B Biointerfaces. 159: 89-96. PMID: 28780464
- Copper(II)-Catalyzed Reactions of α-Keto Thioesters with Azides via C-C and C-S Bond Cleavages: Synthesis of N-Acylureas and Amides. | Maity, R., et al. 2018. J Org Chem. 83: 2114-2124. PMID: 29393648
- Synthesis and biological evaluation of semi-synthetic albocycline analogs. | Daher, SS., et al. 2020. Bioorg Med Chem Lett. 30: 127509. PMID: 32827630
- Transition-metal-free amination phosphoryl azide for the synthesis of phosphoramidates. | Li, Q., et al. 2019. RSC Adv. 9: 16040-16043. PMID: 35521381
- Characterizing Three Azides for Their Potential Use as C. elegans Anesthetics. | Tu, S., et al. 2023. MicroPubl Biol. 2023: PMID: 37082349
- SRS-A antagonist pyranoquinolone alkaloids from east African Fagara plants and their synthesis. | Kamikawa, T., et al. 1996. Bioorg Med Chem. 4: 1317-20. PMID: 8879553
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Diphenyl phosphoryl azide, 5 g | sc-255113 | 5 g | $24.00 | |||
Diphenyl phosphoryl azide, 25 g | sc-255113A | 25 g | $70.00 |
