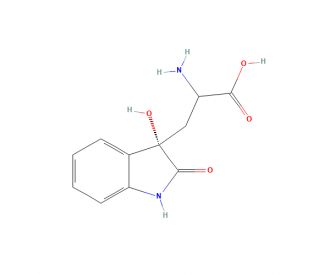
Molecular structure of Dioxindolyl-L-alanine, CAS Number: 184955-21-3
Dioxindolyl-L-alanine (CAS 184955-21-3)
Alternate Names:
(αS)-α-Amino-2,3-dihydro-3-hydroxy-2-oxo-1H-indole-3-propanoic Acid
CAS Number:
184955-21-3
Molecular Weight:
236.22
Molecular Formula:
C11H12N2O4
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Dioxindolyl-L-alanine plays a role in the field of peptide synthesis, particularly in the creation of peptides enriched with unique amino acids such as 5-Hydroxytryptophan, Oxindolylalanine, N-Formylkynurenine, and Kynurenine. Its utility stems from its ability to introduce these specialized amino acids into peptide chains, thereby enabling researchers to explore the biochemical pathways and mechanisms in which these amino acids participate. Specifically, the inclusion of Dioxindolyl-L-alanine in peptide synthesis facilitates the study of tryptophan metabolism and its downstream effects, which are pivotal in understanding various biological processes and responses.
Dioxindolyl-L-alanine (CAS 184955-21-3) References
- Indole can act as an extracellular signal to regulate biofilm formation of Escherichia coli and other indole-producing bacteria. | Martino, PD., et al. 2003. Can J Microbiol. 49: 443-9. PMID: 14569285
- Mechanism of binding of substrate analogues to tryptophan indole-lyase: studies using rapid-scanning and single-wavelength stopped-flow spectrophotometry. | Phillips, RS., et al. 1990. Biochemistry. 29: 8608-14. PMID: 2271544
- A straightforward kinetic evidence for coexistence of 'induced fit' and 'selected fit' in the reaction mechanism of a mutant tryptophan indole lyase Y72F from Proteus vulgaris. | Faleev, NG., et al. 2014. Biochim Biophys Acta. 1844: 1860-7. PMID: 25084024
- The crystal structure of Proteus vulgaris tryptophan indole-lyase complexed with oxindolyl-L-alanine: implications for the reaction mechanism. | Phillips, RS., et al. 2018. Acta Crystallogr D Struct Biol. 74: 748-759. PMID: 30082510
- Detection and identification of transient intermediates in the reactions of tryptophan synthase with oxindolyl-L-alanine and 2,3-dihydro-L-tryptophan. Evidence for a tetrahedral (gem-diamine) intermediate. | Roy, M., et al. 1988. Biochemistry. 27: 8661-9. PMID: 3064816
- Structural Basis of the Stereochemistry of Inhibition of Tryptophan Synthase by Tryptophan and Derivatives. | Phillips, RS. and Harris, AP. 2021. Biochemistry. 60: 231-244. PMID: 33428374
- Differential inhibition of tryptophan synthase and of tryptophanase by the two diastereoisomers of 2,3-dihydro-L-tryptophan. Implications for the stereochemistry of the reaction intermediates. | Phillips, RS., et al. 1985. J Biol Chem. 260: 14665-70. PMID: 3902836
- Interactions of tryptophan synthase, tryptophanase, and pyridoxal phosphate with oxindolyl-L-alanine and 2,3-dihydro-L-tryptophan: support for an indolenine intermediate in tryptophan metabolism. | Phillips, RS., et al. 1984. Biochemistry. 23: 6228-34. PMID: 6395894
- Tryptophanase from Escherichia coli: catalytic and spectral properties in water-organic solvents. | Faleev, NG., et al. 1994. Biochem Mol Biol Int. 34: 209-16. PMID: 7849621
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Dioxindolyl-L-alanine, 50 mg | sc-497015 | 50 mg | $434.00 |
