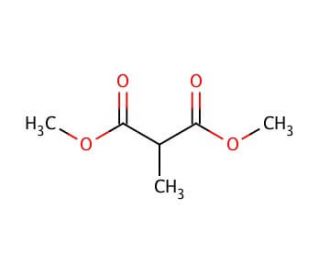
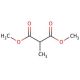
Molecular structure of Dimethyl methylmalonate, CAS Number: 609-02-9
Dimethyl methylmalonate (CAS 609-02-9)
Application:
Dimethyl methylmalonate is a useful biochemical for proteomics research
CAS Number:
609-02-9
Purity:
≥98%
Molecular Weight:
146.14
Molecular Formula:
C6H10O4
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Dimethyl methylmalonate is a versatile compound applied in chemical industries for various purposes. It serves as a key component in the synthesis of fragrances and other organic compounds. Additionally, Dimethyl methylmalonate has demonstrated biochemical and physiological impacts. Though the precise mechanism of action remains incompletely elucidated, it is hypothesized to operate by inhibiting the production of inflammatory mediators, including cytokines and prostaglandins.
Dimethyl methylmalonate (CAS 609-02-9) References
- Solvent-dependent dynamic kinetic asymmetric transformation/kinetic resolution in molybdenum-catalyzed asymmetric allylic alkylations. | Hughes, DL., et al. 2002. J Org Chem. 67: 2762-8. PMID: 11975526
- Formation of a quaternary carbon center through the Pd(0)/PhCOOH-catalyzed allylation of cyclic beta-keto esters and 1,3-diketones with alkynes. | Patil, NT. and Yamamoto, Y. 2004. J Org Chem. 69: 6478-81. PMID: 15357613
- Palladium-catalyzed asymmetric allylic alpha-alkylation of acyclic ketones. | Trost, BM. and Xu, J. 2005. J Am Chem Soc. 127: 17180-1. PMID: 16332054
- Discovery of low nanomolar non-hydroxamate inhibitors of tumor necrosis factor-alpha converting enzyme (TACE). | Duan, JJ., et al. 2007. Bioorg Med Chem Lett. 17: 266-71. PMID: 17027261
- A divergent approach to the preparation of cysteine and serine analogs. | Masterson, DS., et al. 2008. J Pept Sci. 14: 1151-62. PMID: 18613284
- Retention of regiochemistry and chirality in the ruthenium catalyzed allylic alkylation of disubstituted allylic esters. | Kawatsura, M., et al. 2011. J Org Chem. 76: 5485-8. PMID: 21608979
- An Unusual Stereochemical Outcome in the Oxidatively Induced Reductive Elimination of (Pentenediyl) iron Complexes | Yun, Y. K., & Donaldson, W. A. 1997. Journal of the American Chemical Society. 119(17): 4084-4085.
- Retention of regiochemistry of allylic esters in palladium-catalyzed allylic alkylation in the presence of a MOP ligand | Hayashi, T., Kawatsura, M., & Uozumi, Y. 1998. Journal of the American Chemical Society. 120(8): 1681-1687.
- Chiral ditopic receptors. Application to palladium-catalyzed allylic alkylation | Bourguignon, J., Bremberg, U., Dupas, G., Hallman, K., Hagberg, L., Hortala, L.,.. & Rahm, F. 2003. Tetrahedron. 59(48): 9583-9589.
- New phosphine–imine ligands derived from d-gluco- and d-galactosamine in Pd-catalysed asymmetric allylic alkylation | Szulc, I., Kołodziuk, R., Kryczka, B., & Zawisza, A. 2015. Tetrahedron letters. 56(33): 4740-4743.
- Iodine-mediated direct α-amination of dimethyl methylmalonate using non-protected amines | Takeda, M., Maejima, S., Yamaguchi, E., & Itoh, A. 2021. Tetrahedron Letters. 77: 153251.
- Nickel-catalyzed hydroalkylation of 1, 3-dienes with malonates using a homoallyl carbonate as the 1, 3-diene and hydride source | Tsuji, H., Takahashi, Y., & Kawatsura, M. 2021. Tetrahedron Letters. 68: 152916.
- Intermolecular direct catalytic cross-Michael/Michael reactions and tandem Michael/Michael/aldol reactions to linear compounds | Y Asaji, H Maruyama, T Yoshimura, J Matsuo. 2022. Tetrahedron. 122: 132951.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Dimethyl methylmalonate, 1 g | sc-234740A | 1 g | $20.00 | |||
Dimethyl methylmalonate, 5 g | sc-234740 | 5 g | $46.00 | |||
Dimethyl methylmalonate, 10 g | sc-234740B | 10 g | $79.00 | |||
Dimethyl methylmalonate, 25 g | sc-234740C | 25 g | $188.00 | |||
Dimethyl methylmalonate, 100 g | sc-234740D | 100 g | $557.00 |
