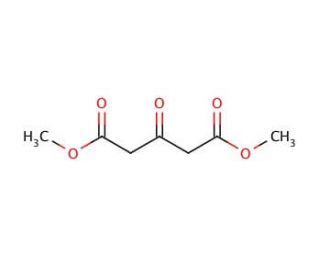
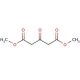
Dimethyl-1,3-acetonedicarboxylate (CAS 1830-54-2)
QUICK LINKS
Dimethyl-1,3-acetonedicarboxylate is a natural compound derived from glucose fermentation by the yeast species Pichia pastoris. This five-carbon dicarboxylic acid contains a methyl ester group at the 3-position, endowing it with remarkable versatility and wide-ranging applications in the chemical industry. As a reagent, it contributes to the formation of compounds like amino acids and peptides. Additionally, its involvement extends to the creation of polymers, surfactants, and other materials. Scientific research has extensively explored the unique structure and properties of Dimethyl-1,3-acetonedicarboxylate, further substantiating its significance. As a participant in synthesizing various organic compounds, such as esters, amides, and nitriles, its importance becomes even more apparent.
Dimethyl-1,3-acetonedicarboxylate (CAS 1830-54-2) References
- Convenient Synthesis of N-Benzyl-1,4-dihydropyridines, Cyclohexenones, and Bicyclo[3.3.1]nonan-3-one Derivatives from 1-Aza-1,3-butadienes. | Geirsson, JK. and Johannesdottir, JF. 1996. J Org Chem. 61: 7320-7325. PMID: 11667657
- Novel hypoglycemic dihydropyridones serendipitously discovered from O- versus C-alkylation in the synthesis of VMAT2 antagonists. | Xie, Y., et al. 2008. Bioorg Med Chem Lett. 18: 5111-4. PMID: 18752945
- Diphenylprolinol silyl ether catalysis in an asymmetric formal carbo [3 + 3] cycloaddition reaction via a domino Michael/Knoevenagel condensation. | Hayashi, Y., et al. 2009. Org Lett. 11: 45-8. PMID: 19053718
- Reactions of 3-acylchromones with dimethyl 1,3-acetonedicarboxylate and 1,3-diphenylacetone: one-pot synthesis of functionalized 2-hydroxybenzophenones, 6H-benzo[c]chromenes and benzo[c]coumarins. | Iaroshenko, VO., et al. 2012. Org Biomol Chem. 10: 9344-8. PMID: 23104252
- Aminobenzosuberone Scaffold as a Modular Chemical Tool for the Inhibition of Therapeutically Relevant M1 Aminopeptidases. | Salomon, E., et al. 2018. Molecules. 23: PMID: 30314342
- Gold(I)-Mediated Cycloisomerization/Cycloaddition Enables Bioinspired Syntheses of Neonectrolides B-E and Analogues. | Purgett, TJ., et al. 2019. J Am Chem Soc. 141: 15135-15144. PMID: 31469554
- Polarized Helical Coumarins: [1,5] Sigmatropic Rearrangement and Excited-State Intramolecular Proton Transfer. | Kielesiński, Ł., et al. 2021. J Org Chem. 86: 6148-6159. PMID: 33830755
- Self-sufficient asymmetric reduction of β-ketoesters catalysed by a novel and robust thermophilic alcohol dehydrogenase co-immobilised with NADH. | Orrego, AH., et al. 2021. Catal Sci Technol. 11: 3217-3230. PMID: 34094502
- Design, synthesis, and in vitro and in vivo characterization of new memantine analogs for Alzheimer's disease. | Turcu, AL., et al. 2022. Eur J Med Chem. 236: 114354. PMID: 35453065
- Chemically Recyclable Poly(β-thioether ester)s Based on Rigid Spirocyclic Ketal Diols Derived from Citric Acid. | Sedrik, R., et al. 2022. Biomacromolecules. 23: 2685-2696. PMID: 35617050
- Development of 1,3-acetonedicarboxylate-derived glucoside amphiphiles (ACAs) for membrane protein study. | Lee, HJ., et al. 2022. Chem Sci. 13: 5750-5759. PMID: 35694361
- Orally efficacious lead of the AVG inhibitor series targeting a dynamic interface in the respiratory syncytial virus polymerase. | Sourimant, J., et al. 2022. Sci Adv. 8: eabo2236. PMID: 35749502
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Dimethyl-1,3-acetonedicarboxylate, 25 g | sc-257362 | 25 g | $33.00 |
