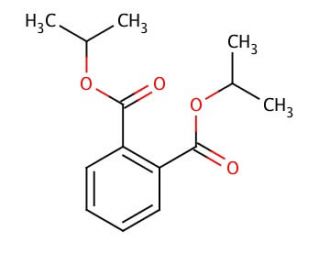
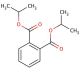
Diisopropyl phthalate (CAS 605-45-8)
QUICK LINKS
Diisopropyl phthalate functions by increasing the flexibility and durability of various polymers and resins, allowing for the production of more malleable and resilient materials. Diisopropyl phthalate interacts at the molecular level by forming strong, non-covalent bonds with the polymer chains, reducing intermolecular forces and increasing the free volume within the material. This results in improved flow properties and reduced brittleness, enhancing the performance of the polymer. Diisopropyl phthalate′s mode of action involves its ability to effectively disperse and separate polymer chains. Diisopropyl phthalate′s function as a plasticizer developments various products, as it enables the production of flexible and durable materials with enhanced mechanical properties.
Diisopropyl phthalate (CAS 605-45-8) References
- Application of sweeping to micellar electrokinetic chromatography-atmospheric pressure chemical ionization-mass spectrometric analysis of environmental pollutants. | Isoo, K., et al. 2001. Electrophoresis. 22: 3426-32. PMID: 11669521
- Evaluation of estrogenic activity of phthalate esters by gene expression profiling using a focused microarray (EstrArray). | Parveen, M., et al. 2008. Environ Toxicol Chem. 27: 1416-25. PMID: 18260696
- Determination of phthalate diesters in foods. | Bradley, EL., et al. 2013. Food Addit Contam Part A Chem Anal Control Expo Risk Assess. 30: 722-34. PMID: 23614781
- Concentrations and distribution of phthalate esters in the seamount area of the Tropical Western Pacific Ocean. | Zhang, Q., et al. 2019. Mar Pollut Bull. 140: 107-115. PMID: 30803624
- Beyond Phthalate Diesters: Existence of Phthalate Monoesters in South China House Dust and Implications for Human Exposure. | Liu, X., et al. 2019. Environ Sci Technol. 53: 11675-11683. PMID: 31503463
- Bis-enolates with Extended π-Conjugation Are Powerful Nucleophiles: A Study of Their Alkylation Reactions with Very Hindered C-Electrophiles. | Castroagudín, M., et al. 2019. J Org Chem. 84: 15805-15816. PMID: 31778066
- Study of atmospheric pressure chemical ionization of phthalates in air by ion mobility spectrometry/mass spectrometry. | Moravský, L., et al. 2021. Rapid Commun Mass Spectrom. 35: e9145. PMID: 34142397
- Stereoselective Synthesis of Hydrindane and Hydroazulene Derivatives by Transannular Cyclization of Nine- and Ten-Membered Carbocycles. | Martínez-García, L., et al. 2021. J Org Chem. 86: 13684-13692. PMID: 34519499
- The reaction laws and toxicity effects of phthalate acid esters (PAEs) ozonation degradation on the troposphere. | Huo, Y., et al. 2022. Environ Pollut. 295: 118692. PMID: 34921942
- Determination of phthalates in bottled waters using solid-phase microextraction and gas chromatography tandem mass spectrometry. | Alshehri, MM., et al. 2022. Chemosphere. 304: 135214. PMID: 35671816
- Enhancement of the mutagenicity of amino acid pyrolysates by phthalate esters. | Sato, T., et al. 1994. Environ Mol Mutagen. 24: 325-31. PMID: 7851345
- Binding characteristics of dialkyl phthalates for the estrogen receptor. | Nakai, M., et al. 1999. Biochem Biophys Res Commun. 254: 311-4. PMID: 9918834
- Vapor Pressures of Diethyl, Diisopropyl, and Dibutyl Phthalates at Reduced Pressures | Katayama Hirotake 1. 1988. Bulletin of the Chemical Society of Japan. Vol.61,: 3326-3328.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Diisopropyl phthalate, 50 ml | sc-234703 | 50 ml | $229.00 |
