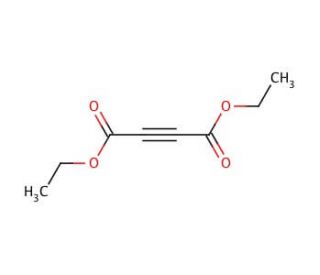
Diethyl acetylenedicarboxylate (CAS 762-21-0)
QUICK LINKS
Diethyl acetylenedicarboxylate is widely used in organic chemistry research, particularly in the synthesis of cyclic and acyclic organic compounds. Its role as a dienophile in Diels-Alder reactions is of special interest as it contributes to the formation of complex molecular structures that are useful in various chemical studies. Furthermore, Diethyl acetylenedicarboxylate is involved in the study of Michael addition reactions, where it acts as an electrophilic component. This compound is also utilized in the synthesis of heterocycles, which are fundamental in the study of chemical reactivity and mechanism. Research involving Diethyl acetylenedicarboxylate helps in advancing the understanding of molecular interactions and reaction pathways, which are critical for the design of new synthetic methodologies.
Diethyl acetylenedicarboxylate (CAS 762-21-0) References
- The surprising nucleophilic addition of aminochlorocarbenes to diethyl acetylenedicarboxylate and to oxalyl chloride: quinolines and benzo[1,4]diazepines from N-alkylformanilides and oxalyl chloride in the presence of Hünig's base. | Cheng, Y., et al. 2003. Org Biomol Chem. 1: 3605-10. PMID: 14599025
- Structure-activity comparison of the cytotoxic properties of diethyl maleate and related molecules: identification of diethyl acetylenedicarboxylate as a thiol cross-linking agent. | West, JD., et al. 2011. Chem Res Toxicol. 24: 81-8. PMID: 21105742
- Unprecedented noncatalyzed anti-carbozincation of diethyl acetylenedicarboxylate through alkylzinc group radical transfer. | Maury, J., et al. 2011. Org Lett. 13: 1884-7. PMID: 21388206
- Enhanced toxicity of the protein cross-linkers divinyl sulfone and diethyl acetylenedicarboxylate in comparison to related monofunctional electrophiles. | West, JD., et al. 2011. Chem Res Toxicol. 24: 1457-9. PMID: 21812477
- Bifunctional electrophiles cross-link thioredoxins with redox relay partners in cells. | Naticchia, MR., et al. 2013. Chem Res Toxicol. 26: 490-7. PMID: 23414292
- Pronounced toxicity differences between homobifunctional protein cross-linkers and analogous monofunctional electrophiles. | Spencer, MK., et al. 2013. Chem Res Toxicol. 26: 1720-9. PMID: 24138115
- Synthesis of novel highly functionalized 4-thiazolidinone derivatives from 4-phenyl-3-thiosemicarbazones. | Benmohammed, A., et al. 2014. Molecules. 19: 3068-83. PMID: 24619352
- 3,4,5-Trisubstituted Furan-2(5H)-one Derivatives: Efficient one-pot Synthesis and Evaluation of Cytotoxic Activity. | Basyouni, WM., et al. 2015. Drug Res (Stuttg). 65: 473-8. PMID: 25207706
- Alkyne activation and polyhedral reorganization in benzothiazolate-capped osmium clusters on reaction with diethyl acetylenedicarboxylate (DEAD) and ethyl propiolate. | Mahid Uddin, K., et al. 2017. Dalton Trans. 46: 13597-13609. PMID: 28952645
- Green Fabrication of Cobalt NPs using Aqueous Extract of Antioxidant Rich Zingiber and Their Catalytic Applications for the Synthesis of Pyrano[2,3-c]pyrazoles. | Mianai, RS., et al. 2019. Comb Chem High Throughput Screen. 22: 18-26. PMID: 30848196
- A Brønsted Acid-Catalyzed Multicomponent Reaction for the Synthesis of Highly Functionalized γ-Lactam Derivatives. | Corte, XD., et al. 2019. Molecules. 24: PMID: 31416281
- Exploration of the Differences between Amine and Thiolate Addition to Acetylenedicarboxylates. | Crumbie, RL., et al. 2019. J Org Chem. 84: 14602-14610. PMID: 31617716
- Substituent-controlled chemoselective synthesis of multi-substituted pyridones via a one-pot three-component cascade reaction. | Liu, S., et al. 2020. Org Biomol Chem. 18: 1130-1134. PMID: 31956881
- Multicomponent Spiropolymerization of Diisocyanides, Diethyl Acetylenedicarboxylate, and Halogenated Quinones. | Zhu, G., et al. 2021. Macromol Rapid Commun. 42: e2100029. PMID: 33987894
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Diethyl acetylenedicarboxylate, 5 g | sc-234652 | 5 g | $23.00 | |||
Diethyl acetylenedicarboxylate, 25 g | sc-234652A | 25 g | $72.00 |
