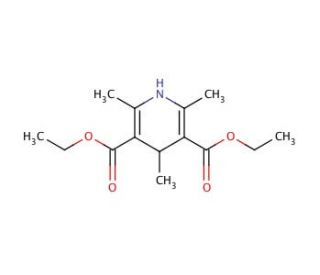
Diethyl 1,4-dihydro-2,4,6-trimethyl-3,5-pyridinedicarboxylate (CAS 632-93-9)
See product citations (1)
QUICK LINKS
Diethyl 1,4-dihydro-2,4,6-trimethyl-3,5-pyridinedicarboxylate blocks heme synthesis and prevents hepatic heme oxygenase-1 induction. It contributes to the formation of Mallory bodies in the liver. Diethyl 1,4-dihydro-2,4,6-trimethyl-3,5-pyridinedicarboxylate plays a role in studying diverse aspects such as enzyme kinetics, metabolic pathways, and cell signaling. Furthermore, it serves as useful for investigating the intricate structure and function of proteins and nucleic acids. Diethyl 1,4-dihydro-2,4,6-trimethyl-3,5-pyridinedicarboxylate effectively targets enzymes like protein kinases and phosphatases by binding to their active sites, impeding substrate binding and hindering enzymatic catalysis. Additionally, Diethyl 1,4-dihydro-2,4,6-trimethyl-3,5-pyridinedicarboxylate exhibits the ability to interact with other proteins and nucleic acids, potentially disrupting their structure and impinging on their functionality.
Diethyl 1,4-dihydro-2,4,6-trimethyl-3,5-pyridinedicarboxylate (CAS 632-93-9) References
- Dexamethasone enhances mallory body formation in drug-primed mouse liver. | Yuan, QX., et al. 2000. Exp Mol Pathol. 69: 202-10. PMID: 11115361
- The role of the ubiquitin-proteasome pathway in the formation of mallory bodies. | Bardag-Gorce, F., et al. 2002. Exp Mol Pathol. 73: 75-83. PMID: 12231209
- Induction of cytochrome P-450 RNA by porphyrogenic agents: on the nature of the coordinate induction with delta-aminolevulinate synthase in tissues of the chicken embryo. | Drew, PD. and Ades, IZ. 1990. Int J Biochem. 22: 607-10. PMID: 1696217
- Transcriptional profiling of bipotential embryonic liver cells to identify liver progenitor cell surface markers. | Ochsner, SA., et al. 2007. Stem Cells. 25: 2476-87. PMID: 17641245
- The regulation of non-coding RNA expression in the liver of mice fed DDC. | Oliva, J., et al. 2009. Exp Mol Pathol. 87: 12-9. PMID: 19362547
- SAMe prevents the up regulation of toll-like receptor signaling in Mallory-Denk body forming hepatocytes. | Bardag-Gorce, F., et al. 2010. Exp Mol Pathol. 88: 376-9. PMID: 20206621
- Sensitivity and specificity of in situ proximity ligation for protein interaction analysis in a model of steatohepatitis with Mallory-Denk bodies. | Zatloukal, B., et al. 2014. PLoS One. 9: e96690. PMID: 24798445
- Regulation of production of delta-aminolaevulinate synthase in tissues of chick embryos. Effects of porphyrogenic agents and of haem precursors. | Drew, PD. and Ades, IZ. 1989. Biochem J. 262: 815-21. PMID: 2590168
- Taurine Attenuates Hepatic Inflammation in Chronic Alcohol-Fed Rats Through Inhibition of TLR4/MyD88 Signaling. | Lin, CJ., et al. 2015. J Med Food. 18: 1291-8. PMID: 26090712
- Affinity purification of antibodies specific for 1,4-dihydropyridine Ca2+ channel blockers. | Sharp, AH. and Campbell, KP. 1987. Circ Res. 61: I37-45. PMID: 3308157
- Lgr5 + cell fate regulation by coordination of metabolic nuclear receptors during liver repair. | Qin, D., et al. 2022. Theranostics. 12: 6130-6142. PMID: 36168631
- Taurocholic acid promotes hepatic stellate cell activation via S1PR2/p38 MAPK/YAP signaling under cholestatic conditions. | Yang, J., et al. 2023. Clin Mol Hepatol. 29: 465-481. PMID: 36800698
- Heat shock in vivo induces Mallory body formation in drug primed mouse liver. | Yuan, QX., et al. 1995. Exp Mol Pathol. 63: 63-76. PMID: 8759055
- Mallory body induction in drug-primed mouse liver. | Yuan, QX., et al. 1996. Hepatology. 24: 603-12. PMID: 8781332
- Mallory body formation by ethanol feeding in drug-primed mice. | Zhang-Gouillon, ZQ., et al. 1998. Hepatology. 27: 116-22. PMID: 9425926
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Diethyl 1,4-dihydro-2,4,6-trimethyl-3,5-pyridinedicarboxylate, 25 g | sc-239721 | 25 g | $151.00 |
