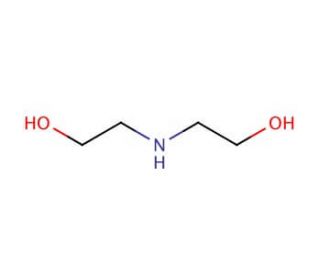
Diethanolamine (CAS 111-42-2)
QUICK LINKS
Diethanolamine (DEA), with the CAS number 111-42-2, is an organic compound characterized by its viscous, hygroscopic, and colorless appearance, though it often appears yellowish due to impurities. Structurally, DEA contains two hydroxyl groups and a secondary amine, making it a versatile chemical in various industrial applications. DEA functions primarily as a surfactant and an emulsifier, facilitating the mixing of hydrophobic and hydrophilic compounds, which is essential in the production of cosmetics, detergents, and shampoos. In research, DEA is extensively used in the study of surfactant systems because it can form complexes with acids, bases, and other compounds, making it useful in understanding phase behavior and molecular interactions in solutions. Its ability to act as an absorption medium for acidic gases makes it significant in environmental chemistry research, particularly in the capture and sequestration of carbon dioxide from industrial emissions, where DEA solutions absorb CO2 during gas processing. Additionally, DEA′s buffer capabilities are utilized in biochemical laboratories for preparing buffer solutions that require specific pH levels for various biochemical and polymerase chain reactions. This wide range of applications highlights DEA′s role in advancing scientific studies by providing essential functionalities in both experimental setups and industrial applications.
Diethanolamine (CAS 111-42-2) References
- Diethanolamine. | . 2000. IARC Monogr Eval Carcinog Risks Hum. 77: 349-79. PMID: 11100407
- Subtyping lymphocytes in peripheral blood by direct immunoalkaline phosphatase labeling and light scatter/absorption flow cytometric analysis. | Kim, YR., et al. 1992. Am J Clin Pathol. 97: 331-7. PMID: 1371900
- Review of the carcinogenic activity of diethanolamine and evidence of choline deficiency as a plausible mode of action. | Leung, HW., et al. 2005. Regul Toxicol Pharmacol. 43: 260-71. PMID: 16188361
- Serum alkaline phosphatase levels in healthy children and evaluation of alkaline phosphatase z-scores in different types of rickets. | Turan, S., et al. 2011. J Clin Res Pediatr Endocrinol. 3: 7-11. PMID: 21448327
- Safety Assessment of Diethanolamine and Its Salts as Used in Cosmetics. | Fiume, MM., et al. 2017. Int J Toxicol. 36: 89S-110S. PMID: 29025348
- Synthesis of symmetrical secondary oligoethylene glycolated amines from diethanolamine. | Yang, H., et al. 2022. Org Biomol Chem. 20: 5129-5138. PMID: 35704908
- Use of Diethanolamine as a Viscous Solvent for Mixture Analysis by Multidimensional Heteronuclear ViscY NMR Experiments. | Leroy, R., et al. 2022. Anal Chem. 94: 9278-9286. PMID: 35737881
- Diethanolamine Modified Perovskite-Substrate Interface for Realizing Efficient ESL-Free PSCs. | Sajid, S., et al. 2023. Nanomaterials (Basel). 13: PMID: 36678003
- Mixed Diethanolamine and Polyethyleneimine with Enhanced CO2 Capture Capacity from Air. | Miao, Y., et al. 2023. Adv Sci (Weinh). 10: e2207253. PMID: 37017566
- Purification of biologically active rubella virus antigens by immunoaffinity chromatography. | Chong, P. and Gillam, S. 1985. J Virol Methods. 10: 261-8. PMID: 3988877
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Diethanolamine, 500 g | sc-211311 | 500 g | $46.00 | |||
Diethanolamine, 1 kg | sc-211311A | 1 kg | $138.00 |
