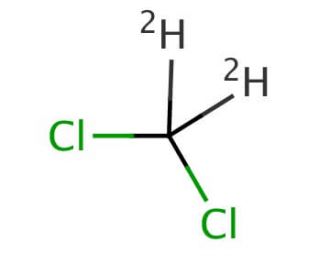
Dichloromethane-d2 (CAS 1665-00-5)
QUICK LINKS
Dichloromethane-d2, also known as deuterated dichloromethane, is a variant of dichloromethane. Deuterated solvents like dichloromethane-d2 are used in nuclear magnetic resonance (NMR) spectroscopy, a powerful analytical technique for determining the structure of organic compounds. The use of deuterated solvents is in NMR because they exhibit significantly lower NMR signal interference compared to their non-deuterated counterparts, thereby providing clearer, more interpretable NMR spectra of the solutes being studied. Similar to regular Dichloromethane, Dichloromethane-d2 has a relatively low boiling point, making it easy to remove from a sample after analysis or reaction by simple evaporation under reduced pressure. Dichloromethane-d2 is chemically stable under a wide range of conditions, making it suitable for use in various chemical reactions and analytical procedures without undergoing significant decomposition or reaction with solutes.
Dichloromethane-d2 (CAS 1665-00-5) References
- Hyperpolarisation of weakly binding N-heterocycles using signal amplification by reversible exchange. | Rayner, PJ., et al. 2021. Chem Sci. 12: 5910-5917. PMID: 34168816
- Heterobimetallic Coinage Metal-Ruthenium Complexes Supported by Anionic N-Heterocyclic Carbenes. | Planer, S., et al. 2021. Chemistry. 27: 15217-15225. PMID: 34342923
- Selective Oxidation of Clopidogrel by Peroxymonosulfate (PMS) and Sodium Halide (NaX) System: An NMR Study. | Krake, EF. and Baumann, W. 2021. Molecules. 26: PMID: 34641465
- Azolium Control of the Osmium-Promoted Aromatic C-H Bond Activation in 1,3-Disubstituted Substrates. | Cancela, L., et al. 2021. Organometallics. 40: 3979-3991. PMID: 34924674
- Diverse synthesis of C2-linked functionalized molecules via molecular glue strategy with acetylene. | Yang, B., et al. 2022. Nat Commun. 13: 1858. PMID: 35388000
- Novel [FeFe]-Hydrogenase Mimics: Unexpected Course of the Reaction of Ferrocenyl α-Thienyl Thioketone with Fe3(CO)12. | Daraosheh, AQ., et al. 2022. Materials (Basel). 15: PMID: 35454560
- Real-Time High-Sensitivity Reaction Monitoring of Important Nitrogen-Cycle Synthons by 15N Hyperpolarized Nuclear Magnetic Resonance. | Rayner, PJ., et al. 2022. J Am Chem Soc. 144: 8756-8769. PMID: 35508182
- Repercussion of a 1,3-Hydrogen Shift in a Hydride-Osmium-Allenylidene Complex. | Esteruelas, MA., et al. 2021. Organometallics. 40: 1523-1537. PMID: 35693112
- Asymmetric Phenyl Substitution: An Effective Strategy to Enhance the Photosensitizing Potential of Curcuminoids. | Vesco, G., et al. 2022. Pharmaceuticals (Basel). 15: PMID: 35890142
- Calix[4]pyrrolato gallate: square planar-coordinated gallium(iii) and its metal-ligand cooperative reactivity with CO2 and alcohols. | Sigmund, LM., et al. 2022. Chem Sci. 13: 11215-11220. PMID: 36320463
- Acetylides for the Preparation of Phosphorescent Iridium(III) Complexes: Iridaoxazoles and Their Transformation into Hydroxycarbenes and N,C(sp3),C(sp2),O-Tetradentate Ligands. | Benítez, M., et al. 2022. Inorg Chem. 61: 19597-19611. PMID: 36416194
- An Organic Molecular Nanobarrel that Hosts and Solubilizes C60. | Bera, S., et al. 2023. Angew Chem Int Ed Engl. 62: e202216540. PMID: 36469042
- Multicolor emission based on a N, N'-Disubstituted dihydrodibenzo [a, c] phenazine crown ether macrocycle. | Ma, CS., et al. 2022. Front Chem. 10: 1087610. PMID: 36545215
- Sensitizer-controlled photochemical reactivity via upconversion of red light. | Glaser, F. and Wenger, OS. 2022. Chem Sci. 14: 149-161. PMID: 36605743
- A molecular descriptor of intramolecular noncovalent interaction for regulating optoelectronic properties of organic semiconductors. | Liu, M., et al. 2023. Nat Commun. 14: 2500. PMID: 37127693
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Dichloromethane-d2, 5 g | sc-257331 | 5 g | $122.00 | |||
Dichloromethane-d2, 10 g | sc-257331A | 10 g | $224.00 |
