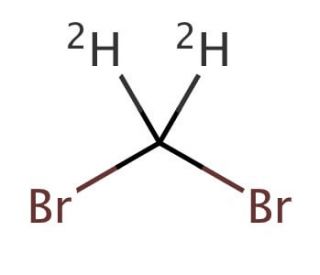
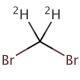
Dibromomethane-d2 (CAS 22117-86-8)
QUICK LINKS
Dibromomethane-d2, also known as deuterated dibromomethane, is a specialized variant of dibromomethane where the hydrogen atoms are replaced with deuterium, a heavier isotope of hydrogen. This modification imbues the molecule with unique properties that are critically valuable in various research applications, particularly in the field of spectroscopy. Deuterated compounds like Dibromomethane-d2 are primarily used in Nuclear Magnetic Resonance (NMR) spectroscopy, where the presence of deuterium reduces the complexity of the NMR spectra. This simplification arises because deuterium has a different magnetic moment compared to hydrogen, which leads to significantly different NMR properties, allowing for clearer and more interpretable spectral data. This is particularly beneficial in complex molecular environments where precise structural elucidation is required. Additionally, Dibromomethane-d2 is utilized in studies of reaction mechanisms involving halogenated methanes. The introduction of deuterium can alter the reaction kinetics due to the kinetic isotope effect, where the stronger C-D bonds compared to C-H bonds influence the rate of bond-breaking and bond-forming steps in chemical reactions. This effect makes Dibromomethane-d2 an invaluable tool in mechanistic studies, providing insights into the subtle dynamics of chemical processes and helping researchers understand how halogenated compounds interact in various chemical environments.
Dibromomethane-d2 (CAS 22117-86-8) References
- PET evaluation of novel radiofluorinated reboxetine analogs as norepinephrine transporter probes in the monkey brain. | Schou, M., et al. 2004. Synapse. 53: 57-67. PMID: 15170818
- Determining volatile organic compounds in human blood from a large sample population by using purge and trap gas chromatography/mass spectrometry. | Ashley, DL., et al. 1992. Anal Chem. 64: 1021-9. PMID: 1590585
- [18F]FMeNER-D2: reliable fully-automated synthesis for visualization of the norepinephrine transporter. | Rami-Mark, C., et al. 2013. Nucl Med Biol. 40: 1049-54. PMID: 24100201
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Dibromomethane-d2, 5 g | sc-227783 | 5 g | $300.00 |
