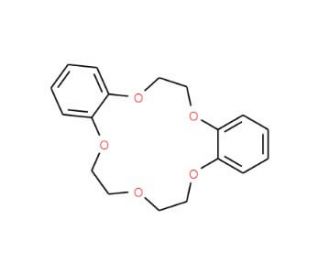
Dibenzo-15-crown-5 (CAS 14262-60-3)
QUICK LINKS
Dibenzo-15-crown-5 (DB15C5) has gained significant prominence in scientific research owing to its exceptional properties as a macrocyclic polyether compound. This cyclic structure is formed by 15 carbon atoms and five oxygen atoms, intricately arranged into a five-membered ring. Dibenzo-15-crown-5 is renowned for its remarkable ability to establish robust complexes with metal ions, serving as a potent chelating agent for a diverse range of metals. Its applications extend to organic synthesis, biochemistry, and materials science, making it an invaluable tool in these fields. The increasing popularity of dibenzo-15-crown-5 within scientific research stems from its distinctive attributes. It finds utility in various realms, including organic synthesis, biochemistry, and materials science. In the domain of organic synthesis, dibenzo-15-crown-5 acts as an effective chelating agent, facilitating the formation of strong complexes with diverse metals. In biochemistry, it has been employed to explore the structure and function of proteins and DNA. Within materials science, dibenzo-15-crown-5 enables investigations into the structure and properties of polymers and other materials. The mechanism of action underlying dibenzo-15-crown-5 revolves around its proficiency in forming robust complexes with metal ions. The macrocyclic structure of dibenzo-15-crown-5 allows for the creation of sturdy complexes with a broad spectrum of metals, encompassing iron, copper, zinc, and magnesium. These complexes are stabilized through the electron-donating properties of the macrocycle′s five oxygen atoms. The electrostatic interactions between the metal ions and the oxygen atoms play a pivotal role in securely holding the metal ions in place.
Dibenzo-15-crown-5 (CAS 14262-60-3) References
- Preparation and decomposition of potassium alkalide-lipophilic crown ether complexes in tetrahydrofuran. | Grobelny, Z., et al. 2002. J Org Chem. 67: 7807-12. PMID: 12398507
- Functional crown ether derivatives as sorbents for ligand exchange chromatography. | Chyueh, SD. and Liu, CY. 1996. Anal Bioanal Chem. 354: 278-83. PMID: 15048448
- Influence of aza crown ethers on the electric percolation of AOT/isooctane/water (w/o) microemulsions. | Dasilva-Carvalhal, J., et al. 2006. J Colloid Interface Sci. 301: 637-43. PMID: 16782122
- Attachment of benzo-crown ethers onto activated carbon cloth to enhance the removal of chromium, cobalt and nickel ions from aqueous solutions by adsorption. | Duman, O. and Ayranci, E. 2010. J Hazard Mater. 176: 231-8. PMID: 19945783
- A comparison of complexation of Li+ ion with macrocyclic ligands 15-crown-5 and 12-crown-4 in binary nitromethane-acetonitrile mixtures by using lithium-7 NMR technique and ab initio calculation. | Alizadeh, N. 2011. Spectrochim Acta A Mol Biomol Spectrosc. 78: 488-93. PMID: 21146449
- Dibenzo crown ether layer formation on muscovite mica. | de Poel, W., et al. 2014. Langmuir. 30: 12570-7. PMID: 25263250
- Supramolecular assembly of isocyanorhodium(I) complexes: an interplay of rhodium(I)···rhodium(I) interactions, hydrophobic-hydrophobic interactions, and host-guest chemistry. | Chan, AK., et al. 2015. J Am Chem Soc. 137: 6920-31. PMID: 25984814
- Geometric and Electronic Structures of Dibenzo-15-Crown-5 Complexes with Alkali Metal Ions Studied by UV Photodissociation and UV-UV Hole-Burning Spectroscopy. | Inokuchi, Y., et al. 2017. J Phys Chem A. 121: 954-962. PMID: 28099814
- Synthesis and interconversions of reduced, alkali-metal supported iron-sulfur-carbonyl complexes. | Shupp, JP., et al. 2017. Dalton Trans. 46: 9163-9171. PMID: 28675227
- Selective Probing of Potassium Ion in Solution by Intramolecular Excimer Fluorescence of Dibenzo-Crown Ethers. | Kida, M., et al. 2018. Chemphyschem. 19: 1331-1335. PMID: 29542235
- Geometric and Electronic Structures of Ag+(benzo-18-crown-6), Ag+(dibenzo-18-crown-6), and Ag+(dibenzo-15-crown-5) Complexes Investigated by Cold Gas-Phase Spectroscopy. | Kitamura, Y., et al. 2019. J Phys Chem A. 123: 9185-9192. PMID: 31545041
- Moment Theory of Chromatography for the Analysis of Reaction Kinetics of Intermolecular Interactions. | Miyabe, K., et al. 2021. Anal Chem. 93: 10365-10371. PMID: 34258992
- Electronic Interaction between Aromatic Chromophores in Dibenzo-Crown Ether Complexes with Alkali Metal Ions Investigated via Cold Gas-Phase Spectroscopy. | Kida, M., et al. 2023. J Phys Chem A. 127: 3210-3220. PMID: 37014846
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Dibenzo-15-crown-5, 1 g | sc-227773 | 1 g | $244.00 |
