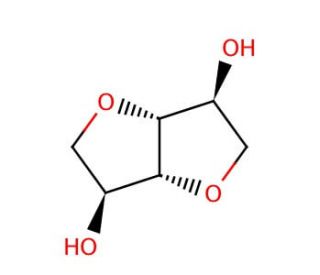
Dianhydro-D-glucitol (CAS 652-67-5)
QUICK LINKS
Dianhydro-D-glucitol, also known as isosorbide, is a bicyclic sugar alcohol derived from sorbitol through intramolecular dehydration. This compound is widely utilized in research due to its rigid structure and stability, which make it an ideal candidate for studying various chemical and biological processes. Dianhydro-D-glucitol′s unique stereochemistry and hydrophobic characteristics enable its use as a monomer in the synthesis of biocompatible and biodegradable polymers. Researchers have explored its application in producing high-performance polyesters and polycarbonates, which are investigated for their mechanical properties, thermal stability, and biodegradability. In carbohydrate chemistry, Dianhydro-D-glucitol serves as a model compound for studying stereoselective reactions and the behavior of cyclic sugar derivatives. Its well-defined structure aids in understanding the conformational aspects and reactivity of similar compounds. Additionally, this chemical is used in developing novel chiral catalysts and ligands for asymmetric synthesis, leveraging its chiral centers to induce enantioselectivity in various reactions. Dianhydro-D-glucitol is also employed in research on osmoprotectants and cryoprotectants, given its ability to stabilize proteins and nucleic acids under stress conditions. By protecting biological samples during freezing and thawing, it facilitates structural and functional studies of biomolecules. Overall, Dianhydro-D-glucitol is a valuable tool in advancing polymer science, carbohydrate chemistry, and biochemistry, providing insights into material properties and stereochemical reactions.
Dianhydro-D-glucitol (CAS 652-67-5) References
- Design of oligolactone-based scaffolds for bone tissue engineering. | Vogt, S., et al. 2005. Biomed Mater Eng. 15: 73-85. PMID: 15623932
- Incorporation of isosorbide into poly(butylene terephthalate) via solid-state polymerization. | Sablong, R., et al. 2008. Biomacromolecules. 9: 3090-7. PMID: 18837541
- Synthesis and applications of ionic liquids derived from natural sugars. | Chiappe, C., et al. 2010. Top Curr Chem. 295: 177-95. PMID: 21626744
- Conversion of cellulose into isosorbide over bifunctional ruthenium nanoparticles supported on niobium phosphate. | Sun, P., et al. 2013. ChemSusChem. 6: 2190-7. PMID: 24115374
- Isosorbide as the structural component of bio-based unsaturated polyesters for use as thermosetting resins. | Sadler, JM., et al. 2014. Carbohydr Polym. 100: 97-106. PMID: 24188843
- Retarder action of isosorbide in a microemulsion for a targeted delivery of ceramide NP into the stratum corneum. | Steinbach, SC., et al. 2017. Pharmazie. 72: 440-446. PMID: 29441901
- Effects of Isosorbide Incorporation into Flexible Polyurethane Foams: Reversible Urethane Linkages and Antioxidant Activity. | Shin, SR., et al. 2019. Molecules. 24: PMID: 30959785
- Investigations of Dianhydro-D-glucitol adsorbed on AuNPs surface: In silico and in vitro approach based on anticancer activity studies against A549 lung cancer cell lines. | V, S., et al. 2021. J Mol Recognit. 34: e2899. PMID: 33783052
- Isosorbide, a versatile green chemical: Elucidating its ADME properties for safe use. | Kim, GB., et al. 2024. Ecotoxicol Environ Saf. 272: 116051. PMID: 38310823
- Acid catalyzed dehydration of alditols. Part I. D-glucitol and D-mannitol. | Bock, Klaus, et al. 1981. Acta Chem Scand. 4: 441-9.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Dianhydro-D-glucitol, 100 g | sc-257316 | 100 g | $57.00 |
