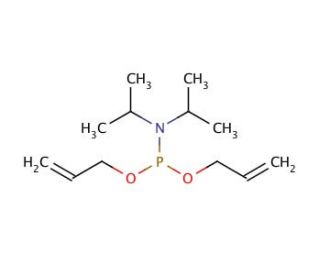
Molecular structure of Diallyl N,N-diisopropylphosphoramidite, CAS Number: 126429-21-8
Diallyl N,N-diisopropylphosphoramidite (CAS 126429-21-8)
CAS Number:
126429-21-8
Purity:
≥90%
Molecular Weight:
245.30
Molecular Formula:
C12H24NO2P
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Diallyl N,N-diisopropylphosphoramidite is a reagent in the synthesis of Leustroducsin B and the synthesis of water-soluble prodrugs of triazole CS-758. Diallyl N,N-diisopropylphosphoramidite is an organophosphorus compound. This colorless liquid with a pungent odor serves as a precursor to phosphoramidites and phosphoramidates. It finds utility as a reagent in synthesizing a range of organic compounds, including peptidomimetics, peptides, and other molecules.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
Alexa Fluor® is a trademark of Molecular Probes Inc., OR., USA
LI-COR® and Odyssey® are registered trademarks of LI-COR Biosciences
Diallyl N,N-diisopropylphosphoramidite (CAS 126429-21-8) References
- Syntheses of glucose derivatives of E5564-related compounds and their LPS-antagonistic activities. | Shiozaki, M., et al. 2006. Carbohydr Res. 341: 811-22. PMID: 16530740
- Synthesis and evaluation of a phosphonate analogue of the soluble guanylate cyclase activator YC-1. | Martin, NI., et al. 2007. Bioorg Med Chem Lett. 17: 4938-41. PMID: 17587571
- A highly convergent synthesis of myristoyl-carba(dethia)-coenzyme A. | Tautz, L. and Rétey, J. 2010. European J Org Chem. 2010: 1728-1735. PMID: 22347809
- Red-emitting rhodamines with hydroxylated, sulfonated, and phosphorylated dye residues and their use in fluorescence nanoscopy. | Kolmakov, K., et al. 2012. Chemistry. 18: 12986-98. PMID: 22968960
- A visualizable chain-terminating inhibitor of glycosaminoglycan biosynthesis in developing zebrafish. | Beahm, BJ., et al. 2014. Angew Chem Int Ed Engl. 53: 3347-52. PMID: 24554559
- Synthesis and SAR studies of benzyl ether derivatives as potent orally active S1P₁ agonists. | Tsuji, T., et al. 2014. Bioorg Med Chem. 22: 4246-56. PMID: 24909680
- Chemical synthesis of Burkholderia Lipid A modified with glycosyl phosphodiester-linked 4-amino-4-deoxy-β-L-arabinose and its immunomodulatory potential. | Hollaus, R., et al. 2015. Chemistry. 21: 4102-14. PMID: 25630448
- Xylosyltransferase II is the predominant isoenzyme which is responsible for the steady-state level of xylosyltransferase activity in human serum. | Kuhn, J., et al. 2015. Biochem Biophys Res Commun. 459: 469-74. PMID: 25748573
- Getting a grip on glycans: A current overview of the metabolic oligosaccharide engineering toolbox. | Sminia, TJ., et al. 2016. Carbohydr Res. 435: 121-141. PMID: 27750120
- Solid-Phase Modular Synthesis of Park Nucleotide and Lipids I and II Analogues. | Katsuyama, A., et al. 2018. Chem Pharm Bull (Tokyo). 66: 84-95. PMID: 29311516
- Total Synthesis and Structural Validation of Phosdiecin A via Asymmetric Alcohol-Mediated Carbonyl Reductive Coupling. | Della-Felice, F., et al. 2019. J Am Chem Soc. 141: 13778-13782. PMID: 31433167
- ArnD is a deformylase involved in polymyxin resistance. | Adak, T., et al. 2020. Chem Commun (Camb). 56: 6830-6833. PMID: 32432293
- Use of Allylic Protecting Groups for the Synthesis of Base-Sensitive Prooligonucleotides | and Nicolas Spinelli, Albert Meyer, Yoshihiro Hayakawa, Jean-Louis Imbach, Jean-Jacques Vasseur. January 2002. EurJOC (European Journal of Organic Chemistry). Volume2002, Issue1: Pages 49-56.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Diallyl N,N-diisopropylphosphoramidite, 1 g | sc-227766 | 1 g | $292.00 |
