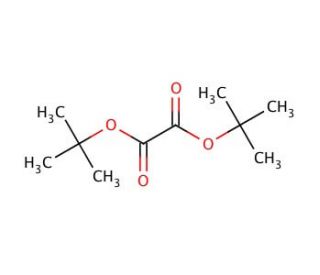
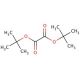
Di-tert-butyl oxalate (CAS 691-64-5)
QUICK LINKS
Di-tert-butyl oxalate is a compound used in synthetic organic chemistry due to its role as a reagent in various organic transformations. Its utility lies in the introduction of the protected oxalate group into organic molecules, which can later undergo decarboxylation or be used as a dienophile in Diels-Alder reactions. Researchers employ di-tert-butyl oxalate to study the kinetics and mechanisms of esterification and transesterification reactions, as its sterically hindered tert-butyl groups provide insight into the effects of steric bulk on reaction rates and outcomes. In addition, the compound is used to generate active carboxylate species under controlled conditions, which is valuable in the study of carboxylation reactions. The tert-butyl groups also render the oxalate ester less prone to hydrolysis, making di-tert-butyl oxalate a stable and versatile reagent for use in a variety of experimental applications.
Di-tert-butyl oxalate (CAS 691-64-5) References
- Perfluoroalkylation with Organosilicon Reagents. | Prakash, GK. and Yudin, AK. 1997. Chem Rev. 97: 757-786. PMID: 11848888
- Highly efficient preparation of aryl beta-diketo acids with tert-butyl methyl oxalate. | Jiang, XH., et al. 2003. J Org Chem. 68: 7555-8. PMID: 12968921
- The presence of long-lived spin states in organic solids with rapid molecular motions. | Fung, BM. and Ermakov, VL. 2004. J Magn Reson. 169: 351-9. PMID: 15261633
- Fluorinated phosphonates: synthesis and biomedical application. | Romanenko, VD. and Kukhar, VP. 2006. Chem Rev. 106: 3868-935. PMID: 16967924
- A platform for designing HIV integrase inhibitors. Part 2: a two-metal binding model as a potential mechanism of HIV integrase inhibitors. | Kawasuji, T., et al. 2006. Bioorg Med Chem. 14: 8420-9. PMID: 17005407
- Crystal packing and melting temperatures of small oxalate esters: the role of C-H···O hydrogen bonding. | Joseph, S., et al. 2011. Acta Crystallogr B. 67: 525-34. PMID: 22101542
- Formation of allylated quaternary carbon centers via C-O/C-O bond fragmentation of oxalates and allyl carbonates. | Chen, H., et al. 2020. Chem Commun (Camb). 56: 454-457. PMID: 31825428
- Cholecystokinin (pancreozymin). 5. Hormonally active desamino derivative of Tyr(SO3H)-Met-Gly-Trp-Met-Asp-Phe-NH2. | Bodanszky, M., et al. 1980. J Med Chem. 23: 82-5. PMID: 6153722
- Potential carcinostatics. 4. Synthesis and biological properties of erythro- and threo-beta-fluoroaspartic acid and erythro-beta-fluoroasparagine. | Wanner, MJ., et al. 1980. J Med Chem. 23: 85-7. PMID: 7359517
- Design and Synthesis of p/m‐[p‐(un)Substituted Phenylsulfonamido]phenyl β‐Diketo Acids and Quinoxalone Derivatives | and Cheng-Chu Zeng, Xue-Mei Li, Hong Yan, Ru-Gang Zhong. 2007. Chinese Journal of Chemistry. 25: 1174-1182.
- Facile synthesis of 2-unsubstituted benzofuran-3-carboxylates using diazo(trimethylsilyl)methylmagnesium bromide | Y Hari, R Kondo, K Date, T Aoyama. 2009. Tetrahedron. 65: 8708-8713.
- Total synthesis of the spirocyclic oxindole alkaloids Corynoxine, Corynoxine B, Corynoxeine, and Rhynchophylline | and Martin J. Wanner, Steen Ingemann, Jan H. van Maarseveen, Henk Hiemstra. 2013. European Journal of Organic Chemistry. 2013: 1100-1106.
- A One-pot Approach to Ethyl 1,4,5-Triaryl-1H-pyrazole-3-carboxylates via an Improved Claisen Condensation-Knorr Reaction Sequence | and Jiaojiao Zhai, Chunhui Gu, Jianan Jiang, Shunli Zhang, Daohua Liao, Lei Wang, Dunru Zhu, Yafei Ji. 2013. Chinese Journal of Chemistry. 31: 1526-1538.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Di-tert-butyl oxalate, 5 g | sc-234536 | 5 g | $90.00 |
