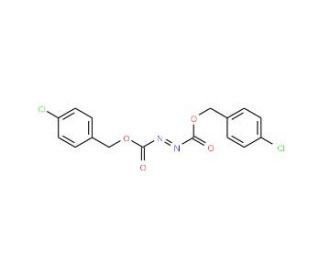
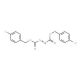
Molecular structure of Di-(4-chlorobenzyl)azodicarboxylate, CAS Number: 916320-82-6
Di-(4-chlorobenzyl)azodicarboxylate (CAS 916320-82-6)
Alternate Names:
Bis(4-chlorobenzyl)azodicarboxylate; DCAD
CAS Number:
916320-82-6
Molecular Weight:
367.18
Molecular Formula:
C16H12Cl2N2O4
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Di-(4-chlorobenzyl)azodicarboxylate (DCAD) is a novel, stable, solid alternative to DEAD and DIAD for a variety of Mitsunobu couplings giving a readily separable hydrazine byproduct that can be recycled.
Di-(4-chlorobenzyl)azodicarboxylate (CAS 916320-82-6) References
- Simplification of the Mitsunobu reaction. Di-p-chlorobenzyl azodicarboxylate: a new azodicarboxylate. | Lipshutz, BH., et al. 2006. Org Lett. 8: 5069-72. PMID: 17048845
- The Mitsunobu reaction: origin, mechanism, improvements, and applications. | But, TY. and Toy, PH. 2007. Chem Asian J. 2: 1340-55. PMID: 17890661
- Mitsunobu and related reactions: advances and applications. | Swamy, KC., et al. 2009. Chem Rev. 109: 2551-651. PMID: 19382806
- N-O tethered carbenoid cyclopropanation facilitates the synthesis of a functionalized cyclopropyl-fused pyrrolidine. | Kalia, D., et al. 2013. J Org Chem. 78: 6131-42. PMID: 23701258
- Cell-Active Small Molecule Inhibitors of the DNA-Damage Repair Enzyme Poly(ADP-ribose) Glycohydrolase (PARG): Discovery and Optimization of Orally Bioavailable Quinazolinedione Sulfonamides. | Waszkowycz, B., et al. 2018. J Med Chem. 61: 10767-10792. PMID: 30403352
- Photochemical Activation of a Hydroxyquinone-Derived Phenyliodonium Ylide by Visible Light: Synthetic and Mechanistic Investigations. | Jalali, M., et al. 2021. J Org Chem. 86: 1758-1768. PMID: 33377772
- Moderately lipophilic 2-(Het)aryl-6-dithioacetals, 2-phenyl-1,4-benzodioxane-6-dithioacetals and 2-phenylbenzofuran-5-dithioacetals: Synthesis and primary evaluation as potential antidiabetic AMPK-activators. | Lepechkin-Zilbermintz, V., et al. 2023. Bioorg Med Chem. 87: 117303. PMID: 37167713
- Azodicarboxylates: Explosive properties and DSC measurements | Berger, A., & Wehrstedt, K. D. 2010. Journal of loss prevention in the process industries. 23(6): 734-739.
- Effect of ring functionalization on the reaction temperature of benzocyclobutene thermoset polymers | Hayes, C. O., Chen, P. H., Thedford, R. P., Ellison, C. J., Dong, G., & Willson, C. G. 2016. Macromolecules. 49(10): 3706-3715.
- Synthetic story of a blockbuster drug: Reboxetine, a potent selective norepinephrine reuptake inhibitor | Shahzad, D., Faisal, M., Rauf, A., & Huang, J. H. 2017. Organic Process Research & Development. 21(11): 1705-1731.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Di-(4-chlorobenzyl)azodicarboxylate, 1 g | sc-300426 | 1 g | $29.00 | |||
Di-(4-chlorobenzyl)azodicarboxylate, 5 g | sc-300426A | 5 g | $62.00 | |||
Di-(4-chlorobenzyl)azodicarboxylate, 25 g | sc-300426B | 25 g | $194.00 | |||
Di-(4-chlorobenzyl)azodicarboxylate, 100 g | sc-300426C | 100 g | $612.00 |
