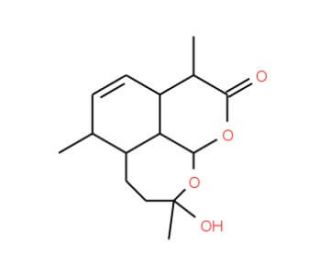
Deoxy Artemisinin (CAS 72826-63-2)
See product citations (1)
QUICK LINKS
Deoxy artemisinin, derived from the naturally occurring compound artemisinin, serves as an important chemical in the domain of scientific research, primarily for its role in biochemical and cellular studies. Unlike its parent compound, deoxy artemisinin lacks an endoperoxide bridge, which significantly alters its chemical behavior and reactivity. This modification is crucial as it affects the compound′s interaction with various molecular targets, enabling researchers to dissect the role of specific functional groups in biological processes. In biochemical research, deoxy artemisinin has been utilized to investigate the mechanistic pathways of artemisinin derivatives. By studying how this compound interacts with proteins and other cellular components, scientists can gain insights into the non-peroxidic mechanisms that might contribute to biological activity. This is particularly relevant in studies aimed at understanding oxidative stress and redox biology, where the absence of the endoperoxide group in deoxy artemisinin allows for the exploration of alternative pathways of action. Additionally, deoxy artemisinin serves as a reference compound in comparative studies, helping to delineate how structural changes influence molecular stability and interaction with biomolecules. Its role in such fundamental research helps in advancing the understanding of chemical biology and the development of novel research tools and methodologies.
Deoxy Artemisinin (CAS 72826-63-2) References
- Identification of the human cytochrome P450 enzymes involved in the in vitro metabolism of artemisinin. | Svensson, US. and Ashton, M. 1999. Br J Clin Pharmacol. 48: 528-35. PMID: 10583023
- Metabolite identification of artemether by data-dependent accurate mass spectrometric analysis using an LTQ-Orbitrap hybrid mass spectrometer in combination with the online hydrogen/deuterium exchange technique. | Liu, T., et al. 2011. Rapid Commun Mass Spectrom. 25: 3303-13. PMID: 22006394
- Dihydroartemisinin inhibits the human erythroid cell differentiation by altering the cell cycle. | Finaurini, S., et al. 2012. Toxicology. 300: 57-66. PMID: 22677887
- A plausible mechanism for the antimalarial activity of artemisinin: A computational approach. | Shandilya, A., et al. 2013. Sci Rep. 3: 2513. PMID: 23985481
- QSAR modeling, docking and ADMET studies for exploration of potential anti-malarial compounds against Plasmodium falciparum. | Qidwai, T. 2016. In Silico Pharmacol. 5: 6. PMID: 28726171
- Inhibition of Human Coronaviruses by Antimalarial Peroxides. | Ghosh, AK., et al. 2021. ACS Infect Dis. 7: 1985-1995. PMID: 33783182
- Determination of artemisinin and its analogs in Artemisia annua extracts by capillary electrophoresis - Mass spectrometry. | Nagy, C., et al. 2021. J Pharm Biomed Anal. 202: 114131. PMID: 34023721
- Isomeric Flavonoids of Artemisia annua (Asterales: Asteraceae) as Insect Growth Inhibitors Against Helicoverpa armigera (Lepidoptera: Noctuidae) | Anshul, N., Bhakuni, R. S., Gaur, R., & Singh, D. 2013. Florida Entomologist. 96(3): 897-903.
- In vitro antibacterial activity of ethanol extract of Artemisia annua and its bioactive fractions against fish pathogens | Soares, M. P., Pinheiro, V. R., Cardoso, I. L., Oliveira, A. D. S. S. D., Sartoratto, A., Ishikawa, M. M.,.. & Sampaio, F. G. 2021. Aquaculture Research. 52(4): 1797-1801.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Deoxy Artemisinin, 10 mg | sc-207521 | 10 mg | $337.00 |
