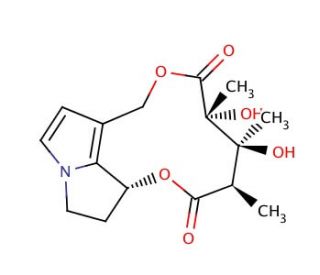
Dehydro Monocrotaline (CAS 23291-96-5)
See product citations (2)
QUICK LINKS
Dehydro monocrotaline is a compound primarily utilized in toxicological research due to its metabolite monocrotaline′s known effects. In research, it serves as an important tool in the study of hepatic veno-occlusive disease and pulmonary hypertension, as it helps in elucidating the biological pathways and cellular responses involved in these conditions. This compound is also used to investigate the metabolic activation and detoxification processes of pyrrolizidine alkaloids, the class of compounds to which dehydro monocrotaline belongs. In addition, it aids in the understanding of the mechanisms of action of these alkaloids at the molecular level, including the formation of DNA adducts and the induction of genotoxicity.
Dehydro Monocrotaline (CAS 23291-96-5) References
- Metabolic activation of pyrrolizidine alkaloids leading to phototoxicity and photogenotoxicity in human HaCaT keratinocytes. | Wang, CC., et al. 2014. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev. 32: 362-84. PMID: 25436474
- Clivorine, an otonecine pyrrolizidine alkaloid from Ligularia species, impairs neuronal differentiation via NGF-induced signaling pathway in cultured PC12 cells. | Xiong, A., et al. 2016. Phytomedicine. 23: 931-8. PMID: 27387401
- Comparative Study of Hepatotoxicity of Pyrrolizidine Alkaloids Retrorsine and Monocrotaline. | Yang, X., et al. 2017. Chem Res Toxicol. 30: 532-539. PMID: 28095673
- Metabolism-mediated cytotoxicity and genotoxicity of pyrrolizidine alkaloids. | He, Y., et al. 2021. Arch Toxicol. 95: 1917-1942. PMID: 34003343
- Constructing vascularized hepatic tissue by cell-assembled viscous tissue sedimentation method and its application for vascular toxicity assessment. | Naito, Y., et al. 2022. Acta Biomater. 140: 275-288. PMID: 34826641
- Inflammation Intensifies Monocrotaline-Induced Liver Injury. | Gong, B., et al. 2023. J Agric Food Chem.. PMID: 36753335
- Dihydropyrrolizine derivatives from unsaturated pyrrolizidine alkaloids. | Mattocks, AR. 1969. J Chem Soc Perkin 1. 8: 1155-62. PMID: 4182485
- Relationship between glutathione concentration and metabolism of the pyrrolizidine alkaloid, monocrotaline, in the isolated, perfused liver. | Yan, CC. and Huxtable, RJ. 1995. Toxicol Appl Pharmacol. 130: 132-9. PMID: 7839360
- Toxicity and carcinogenicity of riddelliine following 13 weeks of treatment to rats and mice. | Chan, PC., et al. 1994. Toxicon. 32: 891-908. PMID: 7985194
- Quantitation of the hepatic release of metabolites of the pyrrolizidine alkaloid, monocrotaline. | Yan, CC. and Huxtable, RJ. 1994. Toxicol Appl Pharmacol. 127: 58-63. PMID: 8048054
- COR pulmonale is caused by monocrotaline and dehydromonocrotaline, but not by glutathione or cysteine conjugates of dihydropyrrolizine. | Pan, LC., et al. 1993. Toxicol Appl Pharmacol. 118: 87-97. PMID: 8430429
- Effect of the pyrrolizidine alkaloid monocrotaline on taurine and sulfur amino acid metabolism in the rat liver. | Yan, CC. and Huxtable, RJ. 1996. Adv Exp Med Biol. 403: 135-45. PMID: 8915351
- Molecular pharmacology and pathophysiological significance of endothelin. | Goto, K., et al. 1996. Jpn J Pharmacol. 72: 261-90. PMID: 9015736
- Involvement of cytochrome P450 3A in the metabolism and covalent binding of 14C-monocrotaline in rat liver microsomes. | Reid, MJ., et al. 1998. J Biochem Mol Toxicol. 12: 157-66. PMID: 9522275
- Effect of taurine on biliary metabolites of glutathione in liver perfused with the pyrrolizidine alkaloid, monocrotaline. | Yan, CC. and Huxtable, RJ. 1998. Adv Exp Med Biol. 442: 85-9. PMID: 9635018
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Dehydro Monocrotaline, 5 mg | sc-214861A | 5 mg | $406.00 | |||
Dehydro Monocrotaline, 25 mg | sc-214861B | 25 mg | $1097.00 | |||
Dehydro Monocrotaline, 50 mg | sc-214861 | 50 mg | $2081.00 |
