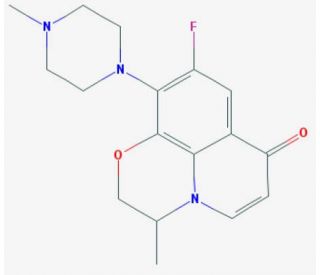
Decarboxyl Ofloxacin (CAS 123155-82-8)
QUICK LINKS
Decarboxyl ofloxacin is a derivative of the fluoroquinolone ofloxacin, where the carboxyl group has been removed. This modification significantly impacts its chemical properties and mechanisms of interaction with biological molecules. Research on decarboxyl ofloxacin has primarily focused on its physicochemical properties, reactivity, and interactions with metal ions in various environments. The mechanism of action of decarboxyl ofloxacin involves the chelation of metal ions. This capability makes it a subject of interest in studies related to metal ion dynamics in biological and environmental systems. The binding of decarboxyl ofloxacin to metals such as magnesium and calcium has been extensively studied using techniques like nuclear magnetic resonance (NMR) spectroscopy and mass spectrometry. These studies help explain how the removal of the carboxyl group affects its binding affinity and specificity compared to its parent compound. Additionally, the chemical stability of decarboxyl ofloxacin under different environmental conditions has been explored. Researchers have employed methods such as high-performance liquid chromatography (HPLC) and thermal analysis to assess its degradation pathways and stability profiles. These investigations are crucial for understanding how structural modifications influence the environmental persistence and reactivity of fluoroquinolone derivatives.
Decarboxyl Ofloxacin (CAS 123155-82-8) References
- Simultaneous determination of multiclass antibiotics and their metabolites in four types of field-grown vegetables. | Tadić, Đ., et al. 2019. Anal Bioanal Chem. 411: 5209-5222. PMID: 31183522
- Elucidating biotransformation pathways of ofloxacin in lettuce (Lactuca sativa L). | Tadić, Đ., et al. 2020. Environ Pollut. 260: 114002. PMID: 31991361
- Occurrence of antibiotics in Lettuce (Lactuca sativa L.) and Radish (Raphanus sativus L.) following organic soil fertilisation under plot-scale conditions: Crop and human health implications. | Matamoros, V., et al. 2022. J Hazard Mater. 436: 129044. PMID: 35525220
- Unveiling the structure–activity relationships of ofloxacin degradation by Co3O4-activated peroxymonosulfate: From microstructures to exposed facets. | Li and Bo, et al. 2023. Chemical Engineering Journal. 467: 143396.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Decarboxyl Ofloxacin, 10 mg | sc-391981 | 10 mg | $360.00 |
