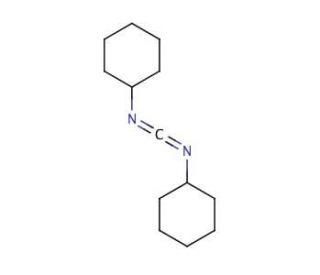
DCC (CAS 538-75-0)
See product citations (3)
QUICK LINKS
DCC, a versatile organic compound, finds widespread use as a coupling reagent in peptide synthesis, making it an invaluable tool in this field. Moreover, it serves as an efficient dehydrating agent, enabling the preparation of various compounds such as amides, esters, nitriles, and anhydrides. In the presence of DCC and dimethyl sulfoxide (DMSO), alcohols can undergo Moffatt oxidation, leading to the conversion of alcohols into aldehydes and ketones. Additionally, it is worth noting that the hydrophobic carbodiimide dicyclohexylcarbodiimide (DCCD) exhibits an intriguing ability to irreversibly inhibit the catalytic subunit of adenosine cyclic 3′,5′-phosphate dependent protein kinase in a time-dependent manner.
DCC (CAS 538-75-0) References
- Isolation of a chloroplast N,N'-dicyclohexylcarbodiimide-binding proteolipid, active in proton translocation. | Nelson, N., et al. 1977. Proc Natl Acad Sci U S A. 74: 2375-8. PMID: 18736
- Covalent modification of the amine transporter with N,N'-dicyclohexylcarbodiimide. | Suchi, R., et al. 1991. Biochemistry. 30: 6490-4. PMID: 2054348
- N,N'-dicyclohexylcarbodiimide is a specific, reversible inhibitor of proline-beta-naphthylamidase. | Takahashi, T. and Takahashi, K. 1990. Biochem Biophys Res Commun. 168: 1001-6. PMID: 2346474
- Single-molecule analysis of F0F1-ATP synthase inhibited by N,N-dicyclohexylcarbodiimide. | Toei, M. and Noji, H. 2013. J Biol Chem. 288: 25717-25726. PMID: 23893417
- Sensitive determination of the potential biomarker sarcosine for prostate cancer by LC-MS with N,N'-dicyclohexylcarbodiimide derivatization. | Chen, J., et al. 2014. J Sep Sci. 37: 14-9. PMID: 24293130
- Synthesis and properties of thermoplastic starch laurates. | Blohm, S. and Heinze, T. 2019. Carbohydr Res. 486: 107833. PMID: 31622867
- Synthesis of amino acid conjugates of glycyrrhizic acid using N-hydroxyphthalimide and N,N'-dicyclohexylcarbodiimide. | Baltina, LA., et al. 2015. Russ J Gen Chem. 85: 2735-2738. PMID: 32288467
- Neutral cyclic sp2-sp3 and sp3-sp3 diboranes from N,N'-dicyclohexylcarbodiimide insertion into 1,2-dichlorodiboranes(4). | Fırıncı, E., et al. 2020. Chem Commun (Camb). 56: 9807-9810. PMID: 32706012
- Electrogenesis in Plasma Membrane Fraction of Halotolerant Microalga Dunaliella maritima and Effects of N,N'-Dicyclohexylcarbodiimide. | Popova, LG., et al. 2020. Biochemistry (Mosc). 85: 930-937. PMID: 33045953
- On the optical purity of peptide active esters prepared by N,N'-dicyclohexylcarbodiimide and 'complexes' of N,N'-dicyclohexylcarbodiimide-pentachlorophenol and N,N'-dicyclohexylcarbodiimide-pentafluorophenol. | Kovacs, J., et al. 1967. J Am Chem Soc. 89: 183-4. PMID: 6040068
- The effect of N,N'-dicyclohexylcarbodiimide on enzymes of bioenergetic relevance. | Azzi, A., et al. 1984. Biochim Biophys Acta. 768: 209-26. PMID: 6095905
- N,N'-dicyclohexylcarbodiimide binds specifically to a single glutamyl residue of the proteolipid subunit of the mitochondrial adenosinetriphosphatases from Neurospora crassa and Saccharomyces cerevisiae. | Sebald, W., et al. 1980. Proc Natl Acad Sci U S A. 77: 785-9. PMID: 6444724
- N,N'-Dicyclohexylcarbodiimide inhibits monovalent cation influx but not cation/proton exchange in heart mitochondria. | Jung, DW., et al. 1980. J Biol Chem. 255: 408-12. PMID: 7356623
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
DCC, 25 g | sc-239713 | 25 g | $72.00 | |||
DCC, 100 g | sc-239713A | 100 g | $208.00 |
