DBCO-maleimide
QUICK LINKS
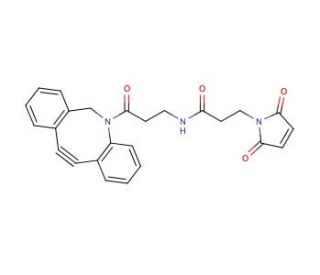
DBCO-maleimide is a specialized chemical linker that integrates the unique properties of dibenzocyclooctyne (DBCO) with the reactivity of a maleimide group, making it a powerful tool in the field of bioconjugation, particularly in bioorthogonal chemistry applications. The DBCO component is renowned for its ability to participate in strain-promoted alkyne-azide cycloaddition (SPAAC), a copper-free click chemistry reaction that facilitates the rapid and specific joining of molecules. This reaction is especially valuable in sensitive biological environments where the introduction of copper could lead to cytotoxic effects. The SPAAC reaction allows for efficient and clean conjugation of azide-bearing molecules at ambient conditions, enhancing the tool′s utility in live-cell labeling and the study of dynamic biological processes. On the other end of the molecule, the maleimide group is specifically reactive towards thiol groups, such as those found on cysteine residues within proteins. This enables precise and stable attachment of proteins, peptides, or other thiol-containing compounds, forming robust thioether bonds without altering the overall structure or function of the biomolecule. The dual functionality of DBCO-maleimide has made it a valuable component in research focused on protein engineering, the development of diagnostic tools, and the assembly of complex molecular architectures. This linker′s ability to bridge molecules with high specificity under physiological conditions supports its widespread use in creating sophisticated biomolecular constructs for advanced research applications.
DBCO-maleimide References
- Site-specific labeling of proteins for electron microscopy. | Dambacher, CM. and Lander, GC. 2015. J Struct Biol. 192: 151-8. PMID: 26409249
- Site-Specific Antibody Labeling by Covalent Photoconjugation of Z Domains Functionalized for Alkyne-Azide Cycloaddition Reactions. | Perols, A., et al. 2015. Chembiochem. 16: 2522-9. PMID: 26417902
- Efficient Formation of Site-Specific Protein-DNA Hybrids Using Copper-Free Click Chemistry. | Mukhortava, A. and Schlierf, M. 2016. Bioconjug Chem. 27: 1559-63. PMID: 27322198
- Targeted Delivery of Cyclotides via Conjugation to a Nanobody. | Kwon, S., et al. 2018. ACS Chem Biol. 13: 2973-2980. PMID: 30248263
- Facile Site-Specific Multiconjugation Strategies in Recombinant Proteins Produced in Bacteria. | Merten, H., et al. 2019. Methods Mol Biol. 2033: 253-273. PMID: 31332759
- Bioorthogonal protein-DNA conjugation methods for force spectroscopy. | Synakewicz, M., et al. 2019. Sci Rep. 9: 13820. PMID: 31554828
- Encapsulating Quantum Dots in Lipid-PEG Micelles and Subsequent Copper-Free Click Chemistry Bioconjugation. | Saeboe, AM., et al. 2020. Methods Mol Biol. 2135: 95-108. PMID: 32246330
- Oligonucleotide conjugated antibody strategies for cyclic immunostaining. | Jones, JA., et al. 2021. Sci Rep. 11: 23844. PMID: 34903759
- Receptor-Targeted Carbon Nanodot Delivery through Polymer Caging and Click Chemistry-Supported LRP1 Ligand Attachment. | Zhang, F., et al. 2023. Polymers (Basel). 15: PMID: 37896282
- Conjugating Hemoglobin and Albumin by Strain-Promoted Azide-Alkyne Cycloaddition. | Lee, C., et al. 2024. Chembiochem. e202400206. PMID: 38837740
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
DBCO-maleimide, 1 mg | sc-397264 | 1 mg | $17.00 | |||
DBCO-maleimide, 5 mg | sc-397264A | 5 mg | $81.00 |
