Davis Oxaziridine (CAS 63160-13-4)
QUICK LINKS
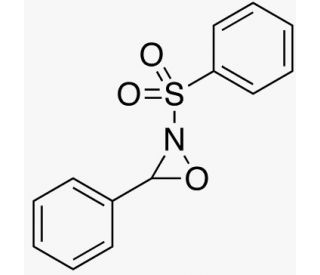
Davis Oxaziridine is an organic compound that is integral to a diverse range of scientific research applications. As a heterocyclic compound, it consists of an oxaziridine ring along with a phenyl ring. Its versatility is showcased through its widespread use in both organic and inorganic synthesis. It acts as a critical intermediate in various reactions, thus facilitating the preparation of other compounds. It finds applications in the synthesis of a myriad of heterocycles, including oxazoles, thiazoles, and additional oxaziridines. Moreover, it plays a role in the creation of pesticides and dyes. While the complete action mechanism of 3-Phenyl-2-(phenylsulfonyl)-1,2-oxaziridine remains not fully deciphered, it is speculated to involve the creation of a cyclic intermediate. This intermediate is then transformed into the desired product through a series of subsequent steps, such as nucleophilic attack, proton transfer, and elimination.
Davis Oxaziridine (CAS 63160-13-4) References
- Oxidation-initiated Nazarov cyclization of vinyl alkoxyallenes. | Spencer, WT., et al. 2011. Org Lett. 13: 414-7. PMID: 21155592
- Illicium sesquiterpenes: divergent synthetic strategy and neurotrophic activity studies. | Trzoss, L., et al. 2013. Chemistry. 19: 6398-408. PMID: 23526661
- A practical synthesis of long-chain iso-fatty acids (iso-C12-C19) and related natural products. | Richardson, MB. and Williams, SJ. 2013. Beilstein J Org Chem. 9: 1807-12. PMID: 24062846
- Total synthesis of viridicatumtoxin B and analogues thereof: strategy evolution, structural revision, and biological evaluation. | Nicolaou, KC., et al. 2014. J Am Chem Soc. 136: 12137-60. PMID: 25317739
- A simple and efficient method for the preparation of 5-hydroxy-3-acyltetramic acids. | Trenner, J. and Prusov, EV. 2015. Beilstein J Org Chem. 11: 323-7. PMID: 25815086
- Synthesis of nonracemic hydroxyglutamic acids. | Piotrowska, DG., et al. 2019. Beilstein J Org Chem. 15: 236-255. PMID: 30745997
- Stereoselective semi-synthesis of the neuroprotective natural product, serofendic acid. | Perusse, D. and Smanski, MJ. 2019. Medchemcomm. 10: 951-960. PMID: 31303993
- Chemical syntheses of the salvinorin chemotype of KOR agonist. | Hill, SJ., et al. 2020. Nat Prod Rep. 37: 1478-1496. PMID: 32808003
- Total Synthesis of Beshanzuenone D and Its Epimers and Abiespiroside A. | Borade, BR., et al. 2020. Org Lett. 22: 8561-8565. PMID: 33104357
- Unified divergent strategy towards the total synthesis of the three sub-classes of hasubanan alkaloids. | Li, G., et al. 2021. Nat Commun. 12: 36. PMID: 33397993
- Evolution of a Strategy for the Total Synthesis of (+)-Cornexistin. | Wildermuth, RE., et al. 2021. Chemistry. 27: 12181-12189. PMID: 34105834
- Synthesis and Evaluation of Marine Natural Product-Inspired Meroterpenoids with Selective Activity toward Dormant Mycobacterium tuberculosis. | Si, A., et al. 2022. ACS Omega. 7: 23487-23496. PMID: 35847331
- Diastereoselective Hydroxylation of N-tert-Butanesulfinyl Imines with 2-(Phenylsulfonyl)-3-phenyloxaziridine (Davis Oxaziridine). | Karmakar, A., et al. 2022. Org Lett. 24: 6548-6553. PMID: 36044766
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Davis Oxaziridine, 1 g | sc-499941 | 1 g | $398.00 |
