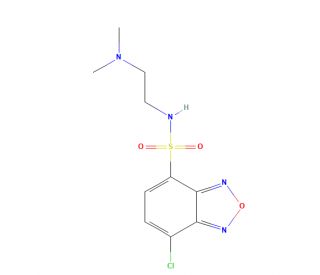
DAABD-Cl (CAS 664985-43-7)
QUICK LINKS
DAABD-Cl is a versatile organic compound utilized both as a reagent in organic synthesis and as a valuable research tool in scientific studies. This white crystalline solid exhibits solubility in various organic solvents, including ethanol, dimethylformamide, and dimethyl sulfoxide. In the realm of organic synthesis, DAABD-Cl finds multiple applications. It is employed in the synthesis of sulfonamides, amines, and amides, contributing to the preparation of diverse compounds. Furthermore, DAABD-Cl serves as a catalyst in the synthesis of heterocyclic compounds such as quinolones, pyridines, and thiazoles. Moreover, DAABD-Cl serves as a valuable research tool in the investigation of biochemical and physiological effects. It has been extensively employed in studying the impact of various drugs on the nervous system and the effects of hormones on the body. Additionally, DAABD-Cl has played a role in examining the influence of environmental pollutants and toxins on the body, as well as the effects of different drugs on the immune and cardiovascular systems. The precise mechanism of action of DAABD-Cl is not yet fully elucidated however, it is hypothesized to function as an enzyme inhibitor, blocking the activity of specific enzymes within the body. Additionally, DAABD-Cl has demonstrated interactions with certain proteins, potentially leading to the inhibition of specific cellular processes. Furthermore, DAABD-Cl exhibits a diverse range of biochemical and physiological effects. It may act to inhibit the activity of enzymes, including cytochrome P450 enzymes involved in drug and chemical metabolism. Furthermore, it has demonstrated the ability to hinder the activity of proteins like cyclooxygenase-2, which is responsible for the production of inflammatory mediators. Additionally, DAABD-Cl has been observed to impede the activity of hormones such as cortisol, for stress response regulation.
DAABD-Cl (CAS 664985-43-7) References
- Fluorogenic derivatization reagents suitable for isolation and identification of cysteine-containing proteins utilizing high-performance liquid chromatography-tandem mass spectrometry. | Masuda, M., et al. 2004. Anal Chem. 76: 728-35. PMID: 14750869
- An improved method for proteomics studies in C. elegans by fluorogenic derivatization, HPLC isolation, enzymatic digestion and liquid chromatography--tandem mass spectrometric identification. | Masuda, M., et al. 2005. Biomed Chromatogr. 19: 556-60. PMID: 15654727
- Synthesis and evaluation of fluorogenic reagents for simultaneous detection of peptides and proteins by HPLC in two different samples. | Saimaru, H., et al. 2006. Biomed Chromatogr. 20: 576-84. PMID: 16779782
- Existence of low-molecular-weight thiols in Caenorhabditis elegans demonstrated by HPLC-fluorescene detection utilizing 7-chloro-N-[2-(dimethylamino)ethyl]-2,1,3-benzoxadiazole-4-sulfonamide. | Asamoto, H., et al. 2007. Biomed Chromatogr. 21: 999-1004. PMID: 17516464
- A proteomics method revealing disease-related proteins in livers of hepatitis-infected mouse model. | Ichibangase, T., et al. 2007. J Proteome Res. 6: 2841-9. PMID: 17559251
- Application of an improved proteomics method, fluorogenic derivatization-liquid chromatography-tandem mass spectrometry, to differential analysis of proteins in small regions of mouse brain. | Asamoto, H., et al. 2008. J Chromatogr A. 1208: 147-55. PMID: 18814880
- Limitation of immunoaffinity column for the removal of abundant proteins from plasma in quantitative plasma proteomics. | Ichibangase, T., et al. 2009. Biomed Chromatogr. 23: 480-7. PMID: 19039805
- Synthesis and evaluation of a fluorogenic reagent for proteomic studies: 7-fluoro-N-[2-(dimethylamino)ethyl]-2,1,3-benzoxadiazole-4-sulfonamide (DAABD-F). | Koshiyama, A. and Imai, K. 2010. Analyst. 135: 2119-24. PMID: 20539882
- FD-LC-MS/MS method for determining protein expression and elucidating biochemical events in tissues and cells. | Ichibangase, T. and Imai, K. 2012. Biol Pharm Bull. 35: 1393-400. PMID: 22975486
- Comprehensive fluorogenic derivatization-liquid chromatography/tandem mass spectrometry proteomic analysis of colorectal cancer cell to identify biomarker candidate. | Koshiyama, A., et al. 2013. Biomed Chromatogr. 27: 440-50. PMID: 22991145
- Efficient chromatographic separation of intact proteins derivatized with a fluorogenic reagent for proteomics analysis. | Ichibangase, T., et al. 2013. Biomed Chromatogr. 27: 1520-3. PMID: 23813469
- Comprehensive and temporal analysis of secreted proteins in the medium from IL-6 exposed human hepatocyte. | Nakata, K., et al. 2014. Biomed Chromatogr. 28: 742-50. PMID: 24861739
- Improved separation of fluorogenic derivatized intact proteins with high resolution and efficiency using a reversed-phase liquid chromatographic system. | Ichibangase, T., et al. 2014. Biomed Chromatogr. 28: 862-7. PMID: 24861755
- Quantification of horse plasma proteins altered by xylazine using the fluorogenic derivatization-liquid chromatography-tandem mass spectrometry. | Mori, M., et al. 2015. J Equine Sci. 26: 141-6. PMID: 26858580
- Structural and functional insights into S-thiolation of human serum albumins. | Nakashima, F., et al. 2018. Sci Rep. 8: 932. PMID: 29343798
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
DAABD-Cl, 100 mg | sc-489828 | 100 mg | $443.00 |
