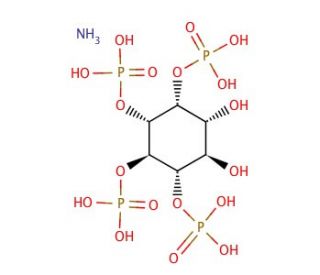
D-myo-Inositol 1,2,5,6-tetrakisphosphate ammonium salt (CAS 91796-88-2)
QUICK LINKS
D-myo-Inositol 1,2,5,6-tetrakisphosphate ammonium salt, a derivative of inositol phosphate, has garnered significant interest in biochemical research due to its crucial roles in cellular signaling and phosphate metabolism. This compound, characterized by phosphate groups at the 1, 2, 5, and 6 positions of the inositol ring, serves as a key intermediate in the phosphoinositide signaling cascade. Research has explaind its mechanism of action, wherein it functions as a substrate for various inositol phosphate kinases and phosphatases, contributing to the generation of important signaling molecules such as inositol pentakisphosphate (IP5) and inositol hexakisphosphate (IP6). Specifically, D-myo-Inositol 1,2,5,6-tetrakisphosphate ammonium salt has been implicated in regulating cellular processes such as calcium mobilization, vesicular trafficking, and gene expression. Furthermore, studies have highlighted its role in modulating ion channel activity, neurotransmitter release, and cytoskeletal dynamics, suggesting its importance in neuronal function and synaptic transmission. Additionally, this compound has been utilized as a molecular probe to dissect the intricacies of inositol phosphate signaling pathways and their implications in various physiological and pathological conditions. Its ability to modulate cellular responses to extracellular stimuli underscores its significance as a versatile research tool in the investigation of molecular mechanisms underlying cellular signaling and phosphate metabolism.
D-myo-Inositol 1,2,5,6-tetrakisphosphate ammonium salt (CAS 91796-88-2) References
- Engineering crop plants: getting a handle on phosphate. | Brinch-Pedersen, H., et al. 2002. Trends Plant Sci. 7: 118-25. PMID: 11906835
- Myo1c binds phosphoinositides through a putative pleckstrin homology domain. | Hokanson, DE., et al. 2006. Mol Biol Cell. 17: 4856-65. PMID: 16971510
- Inositol 1,3,4,5,6-pentakisphosphate 2-kinase from maize: molecular and biochemical characterization. | Sun, Y., et al. 2007. Plant Physiol. 144: 1278-91. PMID: 17535825
- Deep Learning Approach for Predicting the Therapeutic Usages of Unani Formulas towards Finding Essential Compounds. | Wijaya, SH., et al. 2023. Life (Basel). 13: PMID: 36836796
- Enteroinvasive bacteria directly activate expression of iNOS and NO production in human colon epithelial cells. | Witthöft, T., et al. 1998. Am J Physiol. 275: G564-71. PMID: 9724270
- Specificity and promiscuity in phosphoinositide binding by pleckstrin homology domains. | Kavran, JM., et al. 1998. J Biol Chem. 273: 30497-508. PMID: 9804818
- Myo-inositol polyphosphate intermediates in the dephosphorylation of phytic acid by acid phosphatase with phytase activity from rice bran | Hayakawa, T., et al. 1990. Agricultural and Biological Chemistry. 54(2): 279-286.
- Production of D-myo-inositol (1, 2, 4, 5, 6) pentakisphosphate using alginate-entrapped recombinant Pantoea agglomerans glucose-1-phosphatase | Greiner, R. 2008. Brazilian Archives of Biology and Technology. 51: 235-246.
- Principal component and partial least square discriminant based analysis of methanol extracts of bark and regenerated bark of Saraca asoca | Gahlaut, A., et al. 2012. Int J Pharm Pharm Sci. 4: 331-5.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
D-myo-Inositol 1,2,5,6-tetrakisphosphate ammonium salt, 50 µg | sc-214815 | 50 µg | $681.00 |
