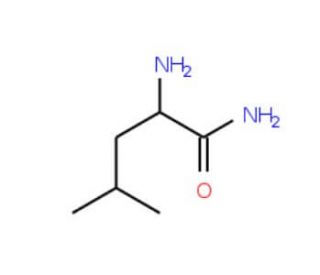
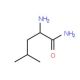
D-Leucine amide (CAS 15893-47-7)
QUICK LINKS
D-Leucine amide is applied in biochemical chemistry, particularly in studies focusing on peptide structure and stability. D-Leucine Amide is instrumental in exploring the properties of peptides that incorporate D-amino acids, which can have implications for understanding protein folding and misfolding. In enzymology, D-Leucine amide serves as a substrate or inhibitor in assays designed to characterize the specificity and mechanism of action of peptidases and other enzymes that interact with D-amino acids. D-Leucine amide is utilized in the development of peptide-based materials, where the incorporation of D-amino acids can enhance stability against enzymatic degradation. D-Leucine amide is being studied in order to understand the role of D-amino acids in cellular signaling and neurological processes.
D-Leucine amide (CAS 15893-47-7) References
- New developments in the synthesis of natural and unnatural amino acids. | Kamphuis, J., et al. 1992. Ann N Y Acad Sci. 672: 510-27. PMID: 1476387
- Purification, characterization, gene cloning and nucleotide sequencing of D: -stereospecific amino acid amidase from soil bacterium: Delftia acidovorans. | Hongpattarakere, T., et al. 2005. J Ind Microbiol Biotechnol. 32: 567-76. PMID: 15959727
- Structure-based organic synthesis of unnatural aeruginosin hybrids as potent inhibitors of thrombin. | Hanessian, S., et al. 2007. Bioorg Med Chem Lett. 17: 3480-5. PMID: 17428662
- New enzymatic method of chiral amino acid synthesis by dynamic kinetic resolution of amino acid amides: use of stereoselective amino acid amidases in the presence of alpha-amino-epsilon-caprolactam racemase. | Yamaguchi, S., et al. 2007. Appl Environ Microbiol. 73: 5370-3. PMID: 17586677
- alpha,alpha-Cyclic aminoacids as useful scaffolds for the preparation of hNK(2) receptor antagonists. | Sisto, A., et al. 2007. Bioorg Med Chem Lett. 17: 4841-4. PMID: 17604625
- A new D-stereospecific amino acid amidase from Ochrobactrum anthropi. | Asano, Y., et al. 1989. Biochem Biophys Res Commun. 162: 470-4. PMID: 2751665
- Properties of a novel D-stereospecific aminopeptidase from Ochrobactrum anthropi. | Asano, Y., et al. 1989. J Biol Chem. 264: 14233-9. PMID: 2760064
- Biocatalytic production of (S)-2-aminobutanamide by a novel d-aminopeptidase from Brucella sp. with high activity and enantioselectivity. | Tang, XL., et al. 2018. J Biotechnol. 266: 20-26. PMID: 29217400
- Microcystins Containing Doubly Homologated Tyrosine Residues from a Microcystis aeruginosa Bloom: Structures and Cytotoxicity. | He, H., et al. 2018. J Nat Prod. 81: 1368-1375. PMID: 29847132
- Bovine-lens leucine aminopeptidase. Kinetic studies with substrates and substrate-like inhibitors. | Fittkau, S., et al. 1974. Eur J Biochem. 44: 523-8. PMID: 4838681
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
D-Leucine amide, 1 g | sc-285370 | 1 g | $66.00 | |||
D-Leucine amide, 5 g | sc-285370A | 5 g | $275.00 |
