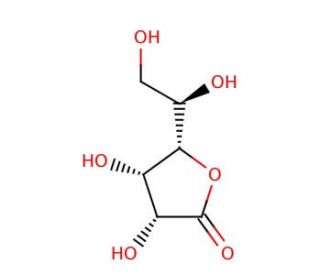
D-Gulono-1,4-lactone (CAS 6322-07-2)
QUICK LINKS
D-Gulono-1,4-lactone, a derivative of the sugar acid D-gulonic acid, plays a critical role in biochemical research due to its involvement in the biosynthesis of ascorbic acid (vitamin C) in animals that are capable of producing it. This compound is particularly important in studies aimed at understanding the biochemical pathways and enzymatic mechanisms that lead to vitamin C synthesis, a pathway absent in humans and certain other animals due to the evolutionary loss of the enzyme L-gulonolactone oxidase. In research applications, D-Gulono-1,4-lactone is extensively used to explore the physiological and molecular biology aspects of ascorbic acid production within cells. It serves as a substrate for the enzyme L-gulonolactone oxidase, which catalyzes the conversion of D-Gulono-1,4-lactone to ascorbic acid. This reaction is a critical step in the ascorbate biosynthesis pathway, making this compound a key tool in studies investigating the genetic and metabolic factors influencing this pathway. Additionally, D-Gulono-1,4-lactone is utilized in comparative biological studies to understand why some species have retained the ability to synthesize vitamin C while others have not. Such research provides insights into evolutionary biology and has implications for understanding disease mechanisms related to oxidative stress and the dietary requirements of different species. This compound′s use in these contexts underscores its value in basic biological research and its contribution to our understanding of vital metabolic processes.
D-Gulono-1,4-lactone (CAS 6322-07-2) References
- Synthesis of L-glucose from D-gulono-1,4-lactone. | Hajkó, J., et al. 1999. Carbohydr Res. 321: 116-20. PMID: 10612005
- Construction and application of a mass spectral and retention time index database generated from plant GC/EI-TOF-MS metabolite profiles. | Wagner, C., et al. 2003. Phytochemistry. 62: 887-900. PMID: 12590116
- Deep Eutectic Solvents Based on Natural Ascorbic Acid Analogues and Choline Chloride. | Maneffa, AJ., et al. 2020. ChemistryOpen. 9: 550-558. PMID: 32373425
- LC-MS-based rheumatoid arthritis serum metabolomics reveals the role of deoxyinosine in attenuating collagen-induced arthritis in mice. | Xu, D., et al. 2024. Heliyon. 10: e30903. PMID: 38778995
- The Gulono-1, 4-Lactones A Review of Their Synthesis, Reactions, and Related Derivatives | Crawford, T. C. 1981. Advances in Carbohydrate Chemistry and Biochemistry. 38: 287-321.
- Conformational studies on aldonolactones by NMR spectroscopy_ conformations of d-glucono-, d-mannono-, d-gulono-and d-galactono-1, 4-lactone in solution | Wałaszek, Z., & Horton, D. 1982. Carbohydrate Research. 105(1): 131-143.
- A new route to some enantiomerically pure substituted morpholines from D-ribono-and D-gulono-1, 4-lactones | Bennis, K., Calinaud, P., Gelas, J., & Ghobsi, M. 1994. Carbohydrate research. 264(1): 33-44.
- Standard molar enthalpies of formation of crystalline stereoisomers of aldono-1, 4-lactones | Flores, H., & Amador, P. 2004. The Journal of Chemical Thermodynamics. 36(11): 1019-1024.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
D-Gulono-1,4-lactone, 10 g | sc-221493 | 10 g | $161.00 |
