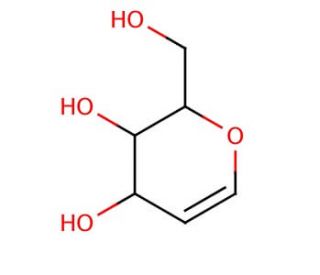
D-Galactal (CAS 21193-75-9)
QUICK LINKS
D-Galactal, a sugar derivative, has garnered attention in glycobiology research due to its role as a precursor in the synthesis of various carbohydrate structures. Its chemical structure, resembling that of a hexose sugar, makes it a key intermediate in the biosynthesis of complex carbohydrates, including glycoproteins, glycolipids, and polysaccharides. Researchers utilize D-Galactal as a substrate in enzymatic assays to study the activity and specificity of glycosyltransferases involved in glycan biosynthesis pathways. By investigating the interactions between D-Galactal and glycosyltransferases, scientists can explain the substrate recognition mechanisms and catalytic mechanisms of these enzymes, contributing to our understanding of glycan diversity and regulation. Moreover, D-Galactal serves as a precursor for the synthesis of glycan mimetics and carbohydrate-based probes, facilitating the study of carbohydrate-mediated processes such as cell adhesion, signaling, and immune recognition. Additionally, D-Galactal finds applications in the development of glycoconjugates, glycomimetics, and carbohydrate-based materials for various biomedical and biotechnological applications, including drug delivery systems, biomaterials, and diagnostics. Overall, D-Galactal plays a crucial role in advancing our understanding of carbohydrate chemistry and glycobiology, offering insights into the roles of carbohydrates in biological processes and providing tools for the development of biomaterials.
D-Galactal (CAS 21193-75-9) References
- The synthesis of 2-deoxy-α-D-glycosides from D-glycals catalyzed by TMSI and PPh3. | Cui, XK., et al. 2012. Carbohydr Res. 358: 19-22. PMID: 22854184
- Unified Synthesis of Right Halves of Halichondrins A-C. | Yahata, K., et al. 2017. J Org Chem. 82: 8792-8807. PMID: 28741352
- Palladium catalyzed stereocontrolled synthesis of C-aryl glycosides using glycals and arenediazonium salts at room temperature. | Singh, AK. and Kandasamy, J. 2018. Org Biomol Chem. 16: 5107-5112. PMID: 29974917
- Palladium(ii)-catalyzed stereoselective synthesis of C-glycosides from glycals with diaryliodonium salts. | Pal, KB., et al. 2020. Org Biomol Chem. 18: 2242-2251. PMID: 32159571
- Concise Synthesis of Tunicamycin V and Discovery of a Cytostatic DPAGT1 Inhibitor. | Mitachi, K., et al. 2022. Angew Chem Int Ed Engl. 61: e202203225. PMID: 35594368
- New glycoconjugation strategies for Ruthenium(II) arene complexes via phosphane ligands and assessment of their antiproliferative activity. | Iacopini, D., et al. 2022. Bioorg Chem. 126: 105901. PMID: 35671646
- Protecting group enabled stereocontrolled approach for rare-sugars talose/gulose via dual-ruthenium catalysis. | Kumar, M., et al. 2023. Carbohydr Res. 523: 108705. PMID: 36370626
- An improved total synthesis of tunicamycin V. | Mitachi, K., et al. 2023. MethodsX. 10: 102095. PMID: 36911209
- Comparison of the beta-galactosidase conformations induced by D-galactal and by magnesium ions. | Viratelle, OM. and Yon, JM. 1980. Biochemistry. 19: 4143-9. PMID: 6774745
- Selective inhibition of metabolic enzymes by enzymatically synthesized D-glucal-6-phosphate. | Chenault, HK. and Mandes, RF. 1994. Bioorg Med Chem. 2: 627-9. PMID: 7858968
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
D-Galactal, 250 mg | sc-221477 | 250 mg | $320.00 |
