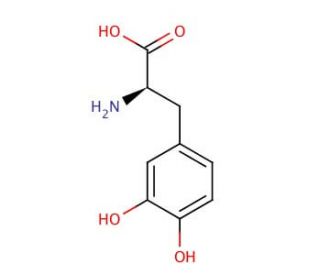
Molecular structure of D-DOPA, CAS Number: 5796-17-8
D-DOPA (CAS 5796-17-8)
Application:
D-DOPA is A DOPA enantiomer that lacks biological activity
CAS Number:
5796-17-8
Molecular Weight:
197.19
Molecular Formula:
C9H11NO4
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
D-DOPA is a DOPA enantiomer that lacks biological activity. D-DOPA is a versatile compound used in numerous scientific research applications. It is used to investigate the structure and function of dopamine receptors, as well as the influence of dopamine in learning and memory processes. D-DOPA is synthesized through enzymatic hydrolysis from L-DOPA, which is derived from the amino acid L-tyrosine. The conversion of L-DOPA to D-DOPA is facilitated by DOPA decarboxylases, which can be found in a variety of organisms including plants, animals, and fungi.
D-DOPA (CAS 5796-17-8) References
- D-dopa is unidirectionally converted to L-dopa by D-amino acid oxidase, followed by dopa transaminase. | Wu, M., et al. 2006. Clin Exp Pharmacol Physiol. 33: 1042-6. PMID: 17042912
- Structural basis of D-DOPA oxidation by D-amino acid oxidase: alternative pathway for dopamine biosynthesis. | Kawazoe, T., et al. 2007. Biochem Biophys Res Commun. 355: 385-91. PMID: 17303072
- Human D-amino acid oxidase: an update and review. | Kawazoe, T., et al. 2007. Chem Rec. 7: 305-15. PMID: 17924443
- Cryptococcus neoformans laccase catalyses melanin synthesis from both D- and L-DOPA. | Eisenman, HC., et al. 2007. Microbiology (Reading). 153: 3954-3962. PMID: 18048910
- D-amino acid oxidase activator gene (DAOA) variation affects cerebrospinal fluid homovanillic acid concentrations in healthy Caucasians. | Andreou, D., et al. 2012. Eur Arch Psychiatry Clin Neurosci. 262: 549-56. PMID: 22454242
- D-dopa and L-dopa similarly elevate brain dopamine and produce turning behavior in rats. | Karoum, F., et al. 1988. Brain Res. 440: 190-4. PMID: 3129126
- Balancing interactions in proline-based receptors for chiral recognition of l-/d-DOPA. | Guo, LE., et al. 2020. Org Biomol Chem. 18: 4590-4598. PMID: 32497164
- Study of tyrosine and dopa enantiomers as tyrosinase substrates initiating l- and d-melanogenesis pathways. | Fernandez-Julia, PJ., et al. 2021. Biotechnol Appl Biochem. 68: 823-831. PMID: 32776353
- D-DOPA Is a Potent, Orally Bioavailable, Allosteric Inhibitor of Glutamate Carboxypeptidase II. | Gori, SS., et al. 2022. Pharmaceutics. 14: PMID: 36297453
- Enzymic sulphation of dopa and tyrosine isomers by HepG2 human hepatoma cells: stereoselectivity and stimulation by Mn2+. | Suiko, M., et al. 1996. Biochem J. 314 (Pt 1): 151-8. PMID: 8660277
- Elevated striatal dopamine levels following administration of D-DOPA and its alpha-keto acid metabolite DHPPA: behavioral and physiological studies in vivo in the rat. | Brannan, T., et al. 1996. Brain Res. 718: 165-8. PMID: 8773780
- Sodium benzoate differentially blocks circling induced by D-and L-dopa in the hemi-parkinsonian rat. | Moses, J., et al. 1996. Neurosci Lett. 218: 145-8. PMID: 8945749
- Comparison of neurotoxicity following repeated administration of l-dopa, d-dopa and dopamine to embryonic mesencephalic dopamine neurons in cultures derived from Fisher 344 and Sprague-Dawley donors. | Alexander, T., et al. 1997. Cell Transplant. 6: 309-15. PMID: 9171163
Activator of:
epsin 3.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
D-DOPA, 500 mg | sc-211173 | 500 mg | $520.00 |
