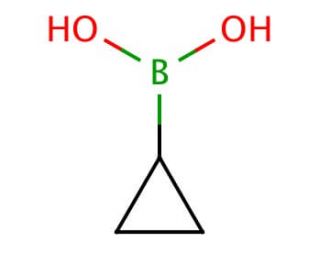
Cyclopropylboronic acid (CAS 411235-57-9)
QUICK LINKS
Cyclopropylboronic acid is primarily utilized in the field of organic chemistry, particularly in Suzuki coupling reactions, which are pivotal for creating biaryl compounds. This chemical is essential for the synthesis of various organic molecules, where the introduction of the cyclopropyl group can significantly alter the properties of the compounds. Additionally, Cyclopropylboronic acid is employed in research focused on the development of new materials with enhanced electronic properties, as the cyclopropyl group can influence the electronic distribution within molecules. Its role extends to the study of molecular interactions in biological systems, offering insights into the stability and reactivity of cyclic structures. Furthermore, Cyclopropylboronic acid is also investigated for its potential in catalytic systems to improve efficiency and selectivity in chemical transformations.
Cyclopropylboronic acid (CAS 411235-57-9) References
- Enantioselective 1,4-addition of cyclopropylboronic acid catalyzed by rhodium/chiral diene complexes. | Takechi, R. and Nishimura, T. 2015. Chem Commun (Camb). 51: 8528-31. PMID: 25892197
- One-Pot, Three-Step Synthesis of Cyclopropylboronic Acid Pinacol Esters from Synthetically Tractable Propargylic Silyl Ethers. | Spencer, JA., et al. 2017. Org Lett. 19: 3891-3894. PMID: 28682080
- Synthesis of aryl cyclopropyl sulfides through copper-promoted S-cyclopropylation of thiophenols using cyclopropylboronic acid. | Benoit, E., et al. 2019. Beilstein J Org Chem. 15: 1162-1171. PMID: 31293663
- Synthesis of Mono-N-Methyl Aromatic Amines from Nitroso Compounds and Methylboronic Acid. | Roscales, S. and Csákÿ, AG. 2019. ACS Omega. 4: 13943-13953. PMID: 31497712
- Diversity-Oriented Enzymatic Synthesis of Cyclopropane Building Blocks. | Wittmann, BJ., et al. 2020. ACS Catal. 10: 7112-7116. PMID: 33282460
- Potassium Trimethylsilanolate Enables Rapid, Homogeneous Suzuki-Miyaura Cross-Coupling of Boronic Esters. | Delaney, CP., et al. 2020. ACS Catal. 10: 73-80. PMID: 33585070
- Reinvestigation of sex pheromone biosynthesis in the moth Trichoplusiani reveals novel quantitative control mechanisms. | Foster, SP. 2022. Insect Biochem Mol Biol. 140: 103700. PMID: 34856351
- 2-(Piperidin-4-yl)acetamides as Potent Inhibitors of Soluble Epoxide Hydrolase with Anti-Inflammatory Activity. | Martín-López, J., et al. 2021. Pharmaceuticals (Basel). 14: PMID: 34959721
- Carba Analogues of Flupirtine and Retigabine with Improved Oxidation Resistance and Reduced Risk of Quinoid Metabolite Formation. | Wurm, KW., et al. 2022. ChemMedChem. 17: e202200262. PMID: 35687532
- Access to Diverse Seleno-spirocyclohexadienones via Ag(II)-Catalyzed Selenylative ipso-Annulation with Se and Boronic Acids. | Raji Reddy, C., et al. 2022. ACS Omega. 7: 38045-38052. PMID: 36312410
- Recent advances in the synthesis and reactivity of MIDA boronates. | Aich, D., et al. 2022. Chem Commun (Camb). 58: 13298-13316. PMID: 36382511
- Synthesis of New Azetidine and Oxetane Amino Acid Derivatives through Aza-Michael Addition of NH-Heterocycles with Methyl 2-(Azetidin- or Oxetan-3-Ylidene)Acetates. | Gudelis, E., et al. 2023. Molecules. 28: PMID: 36770762
- Identification of abemaciclib derivatives targeting cyclin-dependent kinase 4 and 6 using molecular dynamics, binding free energy calculation, synthesis, and pharmacological evaluation. | Zhou, Y., et al. 2023. Front Pharmacol. 14: 1154654. PMID: 37234717
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Cyclopropylboronic acid, 1 g | sc-255044 | 1 g | $72.00 |
