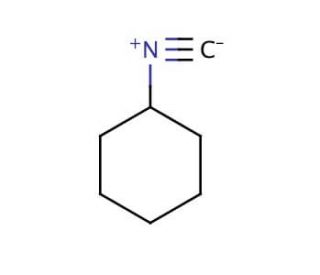
Molecular structure of Cyclohexyl isocyanide, CAS Number: 931-53-3
Cyclohexyl isocyanide (CAS 931-53-3)
Alternate Names:
Isocyanocyclohexane
CAS Number:
931-53-3
Molecular Weight:
109.17
Molecular Formula:
C7H11N
Supplemental Information:
This is as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Cyclohexyl isocyanide (CHIC) is an organic compound belonging to the class of isocyanides that is widely used in the synthesis of agrochemicals, polymers, dyes, organometallic compounds, and optically active compounds. It has also been employed in the synthesis of peptides, proteins, and other biomolecules, as well as in the synthesis of inorganic compounds. Cyclohexyl isocyanide reacts with a variety of organic compounds, including amines, alcohols, and aldehydes, to form amides, esters, cyclic ethers, and lactones. It is also known that this compound can interact with certain enzymes, such as cytochrome P450 enzymes, and disrupt their normal functioning.
Cyclohexyl isocyanide (CAS 931-53-3) References
- Ethylene-like activity of isocyanides. | Quinn, JM. and Yang, SF. 1989. Plant Physiol. 91: 669-73. PMID: 16667085
- Platinum(II) tetramesityltetraphosphane-1,4-diides. | Gómez-Ruiz, S., et al. 2009. Dalton Trans. 2915-20. PMID: 19352518
- Novel isonitrile hydratase involved in isonitrile metabolism. | Sato, H., et al. 2010. J Biol Chem. 285: 34793-802. PMID: 20826798
- Imidazo[1,2-a]pyridin-3-amines as potential HIV-1 non-nucleoside reverse transcriptase inhibitors. | Bode, ML., et al. 2011. Bioorg Med Chem. 19: 4227-37. PMID: 21700466
- Synthesis and cytotoxic activity of novel poly-substituted imidazo[2,1-c][1,2,4]triazin-6-amines. | Akbarzadeh, T., et al. 2015. Mol Divers. 19: 273-81. PMID: 25613858
- Reagents with a Crystalline Coat. | Schwenger, A., et al. 2016. Angew Chem Int Ed Engl. 55: 13706-13709. PMID: 27557595
- Unusual Reactivity of Sodium Tetramesityltetraphosphanediide towards Cyclohexyl Isocyanide. | Adhikari, AK., et al. 2016. Chemistry. 22: 15664-15668. PMID: 27573191
- Rhenium(I) Tricarbonyl Complexes with (2-Hydroxyphenyl)diphenylphosphine as PO Bidentate Ligand. | Shegani, A., et al. 2017. Inorg Chem. 56: 8175-8186. PMID: 28657295
- Synthesis, molecular docking, antimicrobial, antioxidant and toxicity assessment of quinoline peptides. | Thangaraj, M., et al. 2018. J Photochem Photobiol B. 178: 287-295. PMID: 29175602
- Covalent immobilization of laccase by one pot three component reaction and its application in the decolorization of textile dyes. | Salami, F., et al. 2018. Int J Biol Macromol. 120: 144-151. PMID: 30125627
- Reaction between Indazole and Pd-Bound Isocyanides-A Theoretical Mechanistic Study. | Casella, G., et al. 2018. Molecules. 23: PMID: 30423833
- An efficient one-pot synthesis of polyphenolic amino acids and evaluation of their radical-scavenging activity. | Monteiro, LS., et al. 2019. Bioorg Chem. 89: 102983. PMID: 31102692
- A p-tert-Tutyldihomooxacalix[4]arene Based Soft Gel for Sustained Drug Release in Water. | Guo, H., et al. 2020. Front Chem. 8: 33. PMID: 32181237
- A silicon-carbonyl complex stable at room temperature. | Ganesamoorthy, C., et al. 2020. Nat Chem. 12: 608-614. PMID: 32313239
- A methanol and protic ionic liquid Ugi multicomponent reaction path to cytotoxic α-phenylacetamido amides. | Al Otaibi, A., et al. 2019. RSC Adv. 9: 7652-7663. PMID: 35521167
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Cyclohexyl isocyanide, 1 g | sc-257272 | 1 g | $39.00 |
