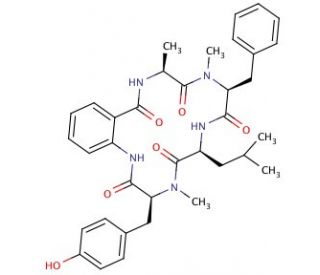
Cycloaspeptide A (CAS 109171-13-3)
QUICK LINKS
Cycloaspeptide A is a cyclic peptide originally isolated from the fungus Aspergillus spp. This compound is characterized by its unique cyclic structure, consisting of several amino acid residues linked in a ring formation, which is crucial for its diverse biological interactions and stability. The structural intricacy of cycloaspeptide A, including both non-standard amino acids and a rare peptide linkage, provides it with a unique three-dimensional conformation. This conformation enables specific interactions with biomolecules, which are of particular interest in biochemical research. One of the primary mechanisms of action studied in cycloaspeptide A is its ability to bind to proteins or enzymes, influencing their activity by allosteric regulation. This interaction is crucial for understanding the dynamics of protein function and enzyme activity at a molecular level. In addition to its biochemical applications, cycloaspeptide A has been used extensively in the study of peptide chemistry. Researchers focus on the synthesis and manipulation of its peptide bonds and ring structure to explore the relationship between molecular structure and stability. These studies are aimed at understanding how modifications to peptide rings can influence biological activity and molecular durability, which is vital for developing new synthetic strategies in peptide chemistry.
Cycloaspeptide A (CAS 109171-13-3) References
- Psychrophilin A and cycloaspeptide D, novel cyclic peptides from the psychrotolerant fungus Penicillium ribeum. | Dalsgaard, PW., et al. 2004. J Nat Prod. 67: 878-81. PMID: 15165155
- Bioactive cyclic peptides from the psychrotolerant fungus Penicillium algidum. | Dalsgaard, PW., et al. 2005. J Antibiot (Tokyo). 58: 141-4. PMID: 15835725
- Four psychrotolerant species with high chemical diversity consistently producing cycloaspeptide A, Penicillium jamesonlandense sp. nov., Penicillium ribium sp. nov., Penicillium soppii and Penicillium lanosum. | Frisvad, JC., et al. 2006. Int J Syst Evol Microbiol. 56: 1427-1437. PMID: 16738124
- Discovery, synthesis, and insecticidal activity of cycloaspeptide E. | Lewer, P., et al. 2006. J Nat Prod. 69: 1506-10. PMID: 17067173
- Cycloaspeptide A and pseurotin A from the endophytic fungus Penicillium janczewskii. | Schmeda-Hirschmann, G., et al. 2008. Z Naturforsch C J Biosci. 63: 383-8. PMID: 18669024
- Cycloaspeptides F and G, Cyclic Pentapeptides from a Cordyceps-colonizing isolate of Isaria farinosa. | Zhang, Y., et al. 2009. J Nat Prod. 72: 1364-7. PMID: 19572613
- Two peptides, cycloaspeptide A and nazumamide A from a sponge associated marine actinobacterium Salinispora sp. | Bose, U., et al. 2014. Nat Prod Commun. 9: 545-6. PMID: 24868880
- Upon DFT-D3 dispersion correction and ECD spectral confirmation, only several conformers can stably coexist for three fungal cycloaspeptides (A, D, G). | Liu, Y., et al. 2022. Spectrochim Acta A Mol Biomol Spectrosc. 283: 121710. PMID: 35952587
- Cycloaspeptide H, a cyclopentapeptide from the endophytic fungus Penicillium virgatum. | Li, BC., et al. 2024. Nat Prod Res. 38: 486-492. PMID: 36190784
- Carboxylate-Directed Pd-Catalyzed β-C(sp3)-H Arylation of N-Methyl Alanine Derivatives for Diversification of Bioactive Peptides. | Yeom, S., et al. 2023. Org Lett. 25: 9008-9013. PMID: 38084750
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Cycloaspeptide A, 1 mg | sc-396522 | 1 mg | $130.00 | |||
Cycloaspeptide A, 5 mg | sc-396522A | 5 mg | $520.00 |
