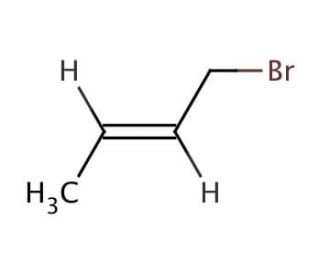
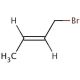
Crotyl bromide (CAS 29576-14-5)
QUICK LINKS
Crotyl bromide an organobromine compound with the chemical formula C4H7Br, finds extensive utility in diverse chemical synthesis processes. Displaying a colorless liquid state, it boasts a boiling point of 109.6°C and a melting point of -41.3°C. This compound exhibits remarkable reactivity and serves as a potent alkylation agent in the realm of organic synthesis, particularly renowned for its role in the creation of agrochemicals, and various other organic entities.In the realm of scientific exploration, crotyl bromide assumes multifaceted roles. Primarily, it acts as a reagent, catalyst, and reactive intermediate within organic synthesis. Its catalytic influence facilitates the synthesis of intricate organic compounds. The exceptionally reactive nature of crotyl bromide enables it to engage in diverse reactions with a spectrum of organic compounds. Its primary application remains centered on alkylation, wherein it effectively reacts with alkenes and alkynes, ultimately giving rise to alkyl halides.
Crotyl bromide (CAS 29576-14-5) References
- Salen as a Chiral Activator: anti versus syn Switchable Diastereoselection in the Enantioselective Addition of Crotyl Bromide to Aromatic Aldehydes We thank the CNR (Rome), M.U.R.S.T. (Rome) 'Progetto Stereoselezione in Chimica Organica, Metodologie ed Applicazioni', and the University of Bologna (funds for selected research topics) for financial support of this research. | Bandini, M., et al. 2000. Angew Chem Int Ed Engl. 39: 2327-2330. PMID: 10941080
- Indium-mediated organometallic reactions in aqueous media. Stereoselectivity in the crotylation of sulfonimines bearing a proximal chelating group. | Lu, W. and Chan, TH. 2001. J Org Chem. 66: 3467-73. PMID: 11348131
- (R)-2,3-O-Cyclohexylideneglyceraldehyde, a Versatile Intermediate for Asymmetric Synthesis of Homoallyl and Homopropargyl Alcohols in Aqueous Medium. | Chattopadhyay, A. 1996. J Org Chem. 61: 6104-6107. PMID: 11667442
- Comparative Diastereoselectivity Analysis of Crotylindium and 3-Bromoallylindium Additions to alpha-Oxy Aldehydes in Aqueous and Nonaqueous Solvent Systems. | Paquette, LA. and Mitzel, TM. 1996. J Org Chem. 61: 8799-8804. PMID: 11667857
- Development of a highly alpha-regioselective metal-mediated allylation reaction in aqueous media: new mechanistic proposal for the origin of alpha-homoallylic alcohols. | Tan, KT., et al. 2003. J Am Chem Soc. 125: 2958-63. PMID: 12617663
- A recyclable electrochemical allylation in water. | Zha, Z., et al. 2005. Org Lett. 7: 1903-5. PMID: 15876015
- Metal nanoparticles or metal oxide nanoparticles, an efficient and promising family of novel heterogeneous catalysts in organic synthesis. | Wang, S., et al. 2009. Dalton Trans. 9363-73. PMID: 19859587
- Indium-mediated asymmetric barbier-type allylations: additions to aldehydes and ketones and mechanistic investigation of the organoindium reagents. | Haddad, TD., et al. 2010. J Org Chem. 75: 642-9. PMID: 20027999
- Synthesis and broad spectrum antiviral evaluation of bis(POM) prodrugs of novel acyclic nucleosides. | Hamada, M., et al. 2013. Eur J Med Chem. 67: 398-408. PMID: 23911854
- Stereoselective Total Synthesis of Bioactive Marine Natural Product Biselyngbyolide B. | Das, S., et al. 2016. Org Lett. 18: 1908-11. PMID: 27043308
- Hemi-Synthesis and Anti-Oomycete Activity of Analogues of Isocordoin. | Escobar, B., et al. 2017. Molecules. 22: PMID: 28604594
- Direct, Mild, and General n-Bu4NBr-Catalyzed Aldehyde Allylsilylation with Allyl Chlorides. | Tekle-Smith, MA., et al. 2017. Org Lett. 19: 6024-6027. PMID: 29068688
- Highly convergent synthesis and antiviral activity of (E)-but-2-enyl nucleoside phosphonoamidates. | Bessières, M., et al. 2018. Eur J Med Chem. 146: 678-686. PMID: 29407990
- Synthesis and Anti-Saprolegnia Activity of New 2',4'-Dihydroxydihydrochalcone Derivatives. | Werner, E., et al. 2020. Antibiotics (Basel). 9: PMID: 32532060
- In(OTf)3-catalyzed intramolecular hydroarylation of α-phenylallyl β-ketosulfones - synthesis of sulfonyl 1-benzosuberones and 1-tetralones. | Chang, MY., et al. 2020. RSC Adv. 10: 18231-18244. PMID: 35517185
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Crotyl bromide, 5 g | sc-255034 | 5 g | $72.00 |
