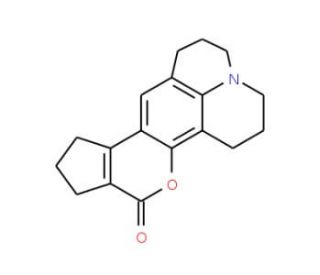
Coumarin 478 (CAS 41175-45-5)
QUICK LINKS
Coumarin 478, also known as Coumarin 106, is a derivative of coumarin, an ester compound, that exhibits anticholinesterase activity in caco-2 cells. It emits fluorescence and can be detected using radiation-emitting tools like x-rays or gamma rays. This fluorescence makes Coumarin 478 particularly valuable in scientific research, where it′s often used as a laser dye or a tracer in fluid dynamics studies due to its ability to emit light at specific wavelengths. Moreover, Coumarin 478′s photostability and high quantum yield make it an excellent choice for applications requiring stable and intense fluorescence. It′s utilized in the development of optical devices, including sensors and bioimaging tools, to track and visualize biological processes with high precision.
Coumarin 478 (CAS 41175-45-5) References
- A rapid screening method for detecting active compounds against erythromycin-resistant bacterial strains of Finnish origin. | Kreander, K., et al. 2005. Folia Microbiol (Praha). 50: 487-93. PMID: 16681145
- Inhibition of acetylcholinesterase by coumarins: the case of coumarin 106. | Fallarero, A., et al. 2008. Pharmacol Res. 58: 215-21. PMID: 18778776
- 2-(4,5,6,7,8,9-Hexahydro-6a-aza-phenyl-en-2-ylmethyl-ene)indan-1,3-dione. | Belyakov, S., et al. 2008. Acta Crystallogr Sect E Struct Rep Online. 64: o1200. PMID: 21202841
- Discovery of dual binding site acetylcholinesterase inhibitors identified by pharmacophore modeling and sequential virtual screening techniques. | Gupta, S., et al. 2011. Bioorg Med Chem Lett. 21: 1105-12. PMID: 21273074
- Determination of ground and excited state dipole moments of dipolar laser dyes by solvatochromic shift method. | Patil, SK., et al. 2014. Spectrochim Acta A Mol Biomol Spectrosc. 123: 117-26. PMID: 24394528
- Molecular docking studies of coumarin hybrids as potential acetylcholinesterase, butyrylcholinesterase, monoamine oxidase A/B and β-amyloid inhibitors for Alzheimer's disease. | Yusufzai, SK., et al. 2018. Chem Cent J. 12: 128. PMID: 30515636
- Coumarin derivatives as acetyl- and butyrylcholinestrase inhibitors: An in vitro, molecular docking, and molecular dynamics simulations study. | Abu-Aisheh, MN., et al. 2019. Heliyon. 5: e01552. PMID: 31183424
- Strategies for Site-Specific Labeling of Receptor Proteins on the Surfaces of Living Cells by Using Genetically Encoded Peptide Tags. | Wolf, P., et al. 2021. Chembiochem. 22: 1717-1732. PMID: 33428317
- Two-photon excitation by the evanescent wave from total internal reflection. | Gryczynski, I., et al. 1997. Anal Biochem. 247: 69-76. PMID: 9126373
- Spatially localized ballistic two-photon excitation in scattering media. | Szmacinski, H., et al. 1998. Biospectroscopy. 4: 303-10. PMID: 9787906
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Coumarin 478, 200 mg | sc-294104 | 200 mg | $412.00 |
