Copper(II) sulfate (CAS 7758-98-7)
See product citations (3)
QUICK LINKS
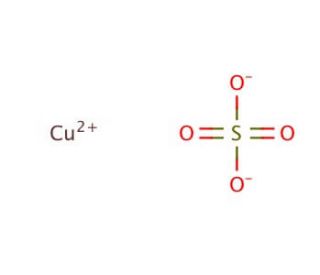
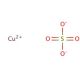
Copper(II) sulfate, also known as cupric sulfate or copper sulphate, is an inorganic compound with the chemical formula CuSO4. It is a blue crystalline solid that is highly soluble in water. Copper(II) sulfate is widely used in various industries and scientific applications. One of the primary uses of copper(II) sulfate is as an agricultural fungicide. It helps control fungal diseases in crops, such as mildew and blight. Additionally, it is used in the manufacturing of Bordeaux mixture, a mixture of copper sulfate and lime, which is a popular fungicidal spray for plants. In the laboratory, copper(II) sulfate is utilized in a range of experiments and demonstrations. It serves as a common reagent for qualitative analysis, allowing for the identification of specific ions in solutions. Copper(II) sulfate is also employed in electroplating processes, where a layer of copper is deposited onto a surface by passing an electric current through a copper sulfate solution. Copper(II) sulfate has other applications as well. In the field of organic synthesis, it can be used as a catalyst or oxidizing agent in certain reactions. It is also employed in the creation of vibrant blue pigments for dyes, paints, and ceramics. In addition, copper(II) sulfate is utilized in the production of copper salts, catalysts, wood preservatives, and animal feed supplements. Copper(II) sulfate plays a significant role in biochemistry research, particularly as a bacteriostatic agent. By binding to specific proteins within the cell, it disrupts vital cellular functions. Its interaction with molecules like DNA and RNA also results in the suppression of gene expression. Consequently, copper sulfate exhibits diverse biochemical and physiological impacts. Notably, Copper(II) sulfate effectively inhibits the growth of bacteria, fungi, and algae. Additionally, it interferes with plant growth by impeding the uptake of essential nutrients. Moreover, Copper(II) sulfate disrupts protein metabolism, leading to the inhibition of critical cellular processes. Its multifaceted properties make it an invaluable tool for studying the intricate mechanisms of biochemistry.
Copper(II) sulfate (CAS 7758-98-7) References
- Effects of copper sulfate and copper nitrate in aquatic medium on the restoration potential and accumulation of copper in stem cuttings of the terrestrial medicinal plant, Portulaca oleracea linn. | Mohanapriya, S., et al. 2006. Environ Monit Assess. 121: 233-44. PMID: 16758284
- Effects of dietary copper (II) sulfate and copper proteinate on performance and blood indexes of copper status in growing pigs. | Feng, J., et al. 2007. Biol Trace Elem Res. 120: 171-8. PMID: 17916969
- In ovo administration of copper nanoparticles and copper sulfate positively influences chicken performance. | Mroczek-Sosnowska, N., et al. 2016. J Sci Food Agric. 96: 3058-62. PMID: 26417698
- Toxic effects of copper sulfate and copper nanoparticles on minerals, enzymes, thyroid hormones and protein fractions of plasma and histopathology in common carp Cyprinus carpio. | Hoseini, SM., et al. 2016. Exp Toxicol Pathol. 68: 493-503. PMID: 27555376
- Copper sulfate pretreatment prevents mitochondrial electron transport chain damage and apoptosis against MPP+-induced neurotoxicity. | Rubio-Osornio, M., et al. 2017. Chem Biol Interact. 271: 1-8. PMID: 28442376
- Transformation of oil palm fronds into pentose sugars using copper (II) sulfate pentahydrate with the assistance of chemical additive. | Loow, YL. and Wu, TY. 2018. J Environ Manage. 216: 192-203. PMID: 28545947
- Degradation reaction of Diazo reactive black 5 dye with copper (II) sulfate catalyst in thermolysis treatment. | Lau, YY., et al. 2018. Environ Sci Pollut Res Int. 25: 7067-7075. PMID: 29275478
- Toxic effects of copper sulfate on diploid and triploid fin cell lines in Misgurnus anguillicaudatus. | Qin, Y., et al. 2018. Sci Total Environ. 643: 1419-1426. PMID: 30189558
- Rutin ameliorates copper sulfate-induced brain damage via antioxidative and anti-inflammatory activities in rats. | Arowoogun, J., et al. 2021. J Biochem Mol Toxicol. 35: e22623. PMID: 32881150
- Impact of copper sulfate on survival, behavior, midgut morphology, and antioxidant activity of Partamona helleri (Apidae: Meliponini). | Bernardes, RC., et al. 2022. Environ Sci Pollut Res Int. 29: 6294-6305. PMID: 34449024
- Inhibition of Oomycetes by the Mixture of Maleic Acid and Copper Sulfate. | Yeon, J., et al. 2022. Plant Dis. 106: 960-965. PMID: 34705489
- How copper can impact pig growth: comparing the effect of copper sulfate and monovalent copper oxide on oxidative status, inflammation, gene abundance, and microbial modulation as potential mechanisms of action. | Forouzandeh, A., et al. 2022. J Anim Sci. 100: PMID: 35723874
- Characterizing the Mechanisms of Metalaxyl, Bronopol and Copper Sulfate against Saprolegnia parasitica Using Modern Transcriptomics. | Wang, Y., et al. 2022. Genes (Basel). 13: PMID: 36140692
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Copper(II) sulfate, 100 g | sc-211133 | 100 g | $46.00 | |||
Copper(II) sulfate, 500 g | sc-211133A | 500 g | $122.00 | |||
Copper(II) sulfate, 1 kg | sc-211133B | 1 kg | $189.00 |
