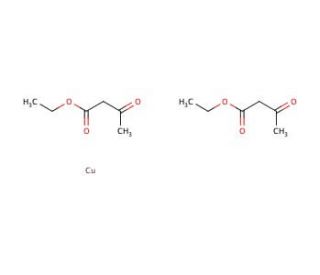
Copper(II) ethylacetoacetate (CAS 14284-06-1)
QUICK LINKS
Copper(II) ethylacetoacetate functions as a catalyst has the formation of carbon-carbon and carbon-heteroatom bonds. It acts as a Lewis acid, facilitating the activation of substrates and promoting the formation of new chemical bonds. The coordination of the copper ion to the carbonyl group of the ethylacetoacetate ligand enhances its reactivity, allowing it to participate in various organic transformations. Through its ability to undergo redox reactions, it can mediate the oxidation of organic compounds, enabling the synthesis of complex molecules. The coordination environment around the copper ion influences its reactivity, making it a versatile function for the construction of diverse chemical structures. In this capacity, Copper(II) ethylacetoacetate plays a role in the development of novel methodologies and the preparation of functional organic materials. Its mechanism of action involves the activation of organic substrates and the modulation of their reactivity, contributing to the advancement of chemical synthesis.
Copper(II) ethylacetoacetate (CAS 14284-06-1) References
- Method for rapid detection of cyanogenic bacteria. | Castric, KF. and Castric, PA. 1983. Appl Environ Microbiol. 45: 701-2. PMID: 16346217
- Precursor to Gas Sensor: A Detailed Study of the Suitability of Copper Complexes as an MOCVD Precursor and their Application in Gas Sensing. | Singh, V., et al. 2021. Inorg Chem. 60: 17141-17150. PMID: 34699217
- New bidentate alkoxy-NHC ligands for enantioselective copper-catalysed conjugate addition | Hervé Clavier, Ludovic Coutable, Jean-Claude Guillemin, Marc Mauduit. 2005. Tetrahedron: Asymmetry. 16: 921-924.
- Mild, base-free copper-catalyzed N-arylations of heterocycles using potassium aryltrifluoroborates in water under air | Nicolas Joubert, Emmanuel Baslé, Michel Vaultier, Mathieu Pucheault. 2010. Tetrahedron Letters. 51: 2994-2997.
- Copper-containing catalysts for solvent-free selective oxidation of benzyl alcohol | Yolanda Pérez, Ruth Ballesteros, Mariano Fajardo, Isabel Sierra, Isabel del Hierro. 2012. Journal of Molecular Catalysis A: Chemical. 32: 45-56.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Copper(II) ethylacetoacetate, 200 mg | sc-268777 | 200 mg | $36.00 |
