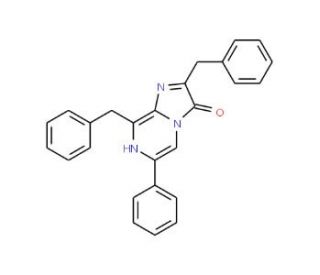
Molecular structure of Coelenterazine 400 a, CAS Number: 70217-82-2
Coelenterazine 400 a (CAS 70217-82-2)
See product citations (3)
Alternate Names:
Coelenterazine 400 a is known as a bisdeoxy derivative of coelenterazine.
Application:
Coelenterazine 400 a is a blue emitting Rluc substrate for bioluminescence resonance energy transfer 2 (BRET2) studies.
CAS Number:
70217-82-2
Purity:
≥95%
Molecular Weight:
391.46
Molecular Formula:
C26H21N3O
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Coelenterazine 400 a is a chromophore that produces the chemiluminescence displayed by the Aequorin protein in the presence of Ca2+. Aequorin is employed as a chemiluminescent probe for intracellular Ca2+, and derivatives of this protein generated with various Coelenterazine derivatives produced a range of Ca2+ probes with varying spectral properties. Coelenterazine 400 a is a bisdeoxy derivative of coelenterazine. It displays an emission maximum of 395nm following conversion by Renilla luciferase (Rluc). Coelenterazine 400 a is paired with class 1 and 3 GFP acceptors.
Coelenterazine 400 a (CAS 70217-82-2) References
- The BRET2/arrestin assay in stable recombinant cells: a platform to screen for compounds that interact with G protein-coupled receptors (GPCRS). | Bertrand, L., et al. 2002. J Recept Signal Transduct Res. 22: 533-41. PMID: 12503639
- Real-time monitoring of receptor and G-protein interactions in living cells. | Galés, C., et al. 2005. Nat Methods. 2: 177-84. PMID: 15782186
- Illuminating insights into protein-protein interactions using bioluminescence resonance energy transfer (BRET). | Pfleger, KD. and Eidne, KA. 2006. Nat Methods. 3: 165-74. PMID: 16489332
- Bioluminescence measurements in mice using a skin window. | Huang, Q., et al. 2007. J Biomed Opt. 12: 054012. PMID: 17994900
- Novel tools for use in bioluminescence resonance energy transfer (BRET) assays. | Robitaille, M., et al. 2009. Methods Mol Biol. 574: 215-34. PMID: 19685312
- Detection of ERalpha-SRC-1 interactions using bioluminescent resonance energy transfer. | Duplessis, TT., et al. 2009. Methods Mol Biol. 590: 253-63. PMID: 19763509
- The chemokine CXC4 and CC2 receptors form homo- and heterooligomers that can engage their signaling G-protein effectors and βarrestin. | Armando, S., et al. 2014. FASEB J. 28: 4509-23. PMID: 25053617
- Sub-nanomolar detection of thrombin activity on a microfluidic chip. | Le, NC., et al. 2014. Biomicrofluidics. 8: 064110. PMID: 25553187
- Bioluminescence of the Ca2+-binding photoprotein aequorin after cysteine modification. | Kurose, K., et al. 1989. Proc Natl Acad Sci U S A. 86: 80-4. PMID: 2643108
- Analysis of Arrestin Recruitment to Chemokine Receptors by Bioluminescence Resonance Energy Transfer. | Bonneterre, J., et al. 2016. Methods Enzymol. 570: 131-53. PMID: 26921945
- Agonist-mediated assembly of the crustacean methyl farnesoate receptor. | Kakaley, EK., et al. 2017. Sci Rep. 7: 45071. PMID: 28322350
- A new inhibitor of the β-arrestin/AP2 endocytic complex reveals interplay between GPCR internalization and signalling. | Beautrait, A., et al. 2017. Nat Commun. 8: 15054. PMID: 28416805
- Insight into the role of urotensin II-related peptide tyrosine residue in UT activation. | Billard, E., et al. 2017. Biochem Pharmacol. 144: 100-107. PMID: 28784291
- Matriptase processing of APLP1 ectodomain alters its homodimerization. | Lanchec, E., et al. 2020. Sci Rep. 10: 10091. PMID: 32572095
- Cause of spectral variation in the luminescence of semisynthetic aequorins. | Shimomura, O. 1995. Biochem J. 306 (Pt 2): 537-43. PMID: 7887908
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Coelenterazine 400 a, 1 mg | sc-280647 | 1 mg | $280.00 | |||
Coelenterazine 400 a, 5 mg | sc-280647A | 5 mg | $790.00 |
