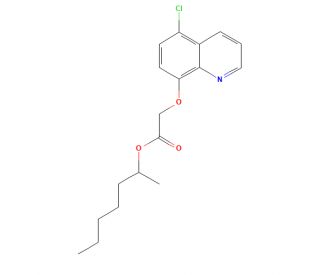
Cloquintocet-Mexyl (CAS 99607-70-2)
QUICK LINKS
Cloquintocet-mexyl (CQMX) is an herbicide created by Dow AgroSciences, and it belongs to the aryloxyphenoxypropionate (AOPP) family of herbicides. As a broad-spectrum, systemic herbicide, Cloquintocet-Mexyl has been utilized to manage a variety of weeds, such as annual and perennial grasses, broadleaf weeds, and sedges. It has proven its effectiveness in controlling a diverse range of weeds across numerous crops, often used in conjunction with other herbicides to enhance its effectiveness. The scientific community has extensively examined cloquintocet-mexyl for its herbicidal efficacy as well as for its potential environmental impact, such as potential contamination of ground and surface water. The mode of action of Cloquintocet-Mexyl involves inhibiting the activity of acetolactate synthase (ALS), an enzyme in the biosynthesis of branched-chain amino acids. Inhibiting ALS results in an accumulation of branched-chain amino acids in the plant, leading to the plant′s eventual demise. Being a systemic herbicide, Cloquintocet-Mexyl is absorbed and transported throughout the plant. It is also photodegradable, breaking down upon exposure to light.
Cloquintocet-Mexyl (CAS 99607-70-2) References
- O-Glucosyltransferase activities toward phenolic natural products and xenobiotics in wheat and herbicide-resistant and herbicide-susceptible black-grass (Alopecurus myosuroides). | Brazier, M., et al. 2002. Phytochemistry. 59: 149-56. PMID: 11809449
- Co-induction of glutathione-S-transferases and multidrug resistance associated protein by xenobiotics in wheat. | Theodoulou, FL., et al. 2003. Pest Manag Sci. 59: 202-14. PMID: 12587874
- Induction of wheat and maize glutathione S-transferase by some herbicide safeners and their effect on enzyme activity against butachlor and terbuthylazine. | Scarponi, L., et al. 2006. Pest Manag Sci. 62: 927-32. PMID: 16835885
- Carboxylesterase activities toward pesticide esters in crops and weeds. | Gershater, M., et al. 2006. Phytochemistry. 67: 2561-7. PMID: 17078983
- Festuca arundinacea, glutathione S-transferase and herbicide safeners: a preliminary case study to reduce herbicidal pollution. | Scarponi, L. and Del Buono, D. 2009. J Environ Sci Health B. 44: 805-9. PMID: 20183093
- The impact of uptake, translocation and metabolism on the differential selectivity between blackgrass and wheat for the herbicide pyroxsulam. | DeBoer, GJ., et al. 2011. Pest Manag Sci. 67: 279-86. PMID: 21104793
- Aryldiones incorporating a [1,4,5]oxadiazepane ring. Part 2: chemistry and biology of the cereal herbicide pinoxaden. | Muehlebach, M., et al. 2011. Pest Manag Sci. 67: 1499-521. PMID: 21656896
- Protective responses induced by herbicide safeners in wheat. | Taylor, VL., et al. 2013. Environ Exp Bot. 88: 93-99. PMID: 23564986
- Cytochrome P450 Inhibitors Reduce Creeping Bentgrass (Agrostis stolonifera) Tolerance to Topramezone. | Elmore, MT., et al. 2015. PLoS One. 10: e0130947. PMID: 26186714
- Metabolism of the14C-labeled herbicide clodinafop-propargyl in plant cell cultures of wheat and tobacco. | Luks, AK., et al. 2016. J Environ Sci Health B. 51: 71-80. PMID: 26578063
- Herbicide Safeners Decrease Sensitivity to Herbicides Inhibiting Acetolactate-Synthase and Likely Activate Non-Target-Site-Based Resistance Pathways in the Major Grass Weed Lolium sp. (Rye-Grass). | Duhoux, A., et al. 2017. Front Plant Sci. 8: 1310. PMID: 28848566
- Suspect and non-target screening of pesticides and pharmaceuticals transformation products in wastewater using QTOF-MS. | Wang, X., et al. 2020. Environ Int. 137: 105599. PMID: 32109725
- Identification of chromosomes in Triticum aestivum possessing genes that confer tolerance to the synthetic auxin herbicide halauxifen-methyl. | Obenland, OA. and Riechers, DE. 2020. Sci Rep. 10: 8713. PMID: 32457385
- Enantioselective separation, analysis and stereoselective dissipation of the chiral pesticide cloquintocet-mexyl using a modified QuEChERS method by high-performance liquid chromatography tandem mass spectrometry. | Li, M., et al. 2022. Chemosphere. 291: 133084. PMID: 34848224
- Simultaneous determination of pinoxaden, cloquintocet-mexyl, clodinafop-propargyl ester and its major metabolite in barley products and soil using QuEChERS modified with multi-walled carbon nanotubes coupled with LC-MS/MS. | Li, N., et al. 2022. Biomed Chromatogr. 36: e5303. PMID: 34957590
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Cloquintocet-Mexyl, 250 mg | sc-491854 | 250 mg | $61.00 |
