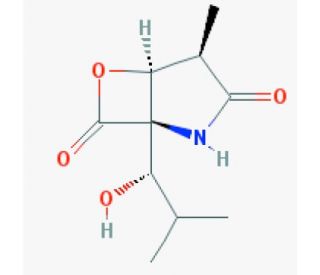
clasto-Lactacystin β-lactone (CAS 155975-72-7)
See product citations (1)
QUICK LINKS
Clasto-Lactacystin β-lactone (CAS: 155975-72-7) is a chemical of significant interest primarily due to its role as a precursor to lactacystin, an intriguing molecule in biochemical research for its ability to modulate proteasome activity. Clasto-Lactacystin β-lactone itself, when metabolized, transforms into lactacystin, which subsequently interacts with the proteasome, a protein complex responsible for degrading unwanted or misfolded proteins in cells. The specific action of clasto-Lactacystin β-lactone involves its conversion to clasto-lactacystin dihydroxyacid in the presence of cellular nucleophiles, which then inhibits the proteasomal activity by specifically targeting the proteolytic β-subunits within the 20S core of the proteasome. This inhibition is crucial for studying protein degradation mechanisms under various physiological conditions, thereby providing insights into the regulation of protein turnover and its implications in cellular functions. Researchers utilize clasto-Lactacystin β-lactone to explore the dynamics of proteasome inhibition and its impact on protein homeostasis within cells.
clasto-Lactacystin β-lactone (CAS 155975-72-7) References
- Neurite outgrowth in PC12 cells. Distinguishing the roles of ubiquitylation and ubiquitin-dependent proteolysis. | Obin, M., et al. 1999. J Biol Chem. 274: 11789-95. PMID: 10206996
- A novel enantioselective synthetic route to omuralide analogues with the potential for species selectivity in proteasome inhibition. | Crane, SN. and Corey, EJ. 2001. Org Lett. 3: 1395-7. PMID: 11348243
- Proteasome inhibitors: from research tools to drug candidates. | Kisselev, AF. and Goldberg, AL. 2001. Chem Biol. 8: 739-58. PMID: 11514224
- Simple enantiospecific syntheses of the C(2)-diastereomers of omuralide and 3-methylomuralide. | Reddy, LR., et al. 2005. Org Lett. 7: 2703-5. PMID: 15957926
- Proteasome inhibition by a totally synthetic beta-lactam related to salinosporamide A and omuralide. | Hogan, PC. and Corey, EJ. 2005. J Am Chem Soc. 127: 15386-7. PMID: 16262399
- Prediction of the mechanism of action of omuralide (clasto-lactacystin beta-lactone) on human cathepsin A based on a structural model of the yeast proteasome beta5/PRE2-subunit/omuralide complex. | Aikawa, S., et al. 2006. Biochim Biophys Acta. 1764: 1372-80. PMID: 16870514
- New entry to convertible isocyanides for the Ugi reaction and its application to the stereocontrolled formal total synthesis of the proteasome inhibitor omuralide. | Gilley, CB., et al. 2007. Org Lett. 9: 3631-4. PMID: 17672474
- Stereospecific total syntheses of proteasome inhibitors omuralide and lactacystin. | Gu, W. and Silverman, RB. 2011. J Org Chem. 76: 8287-93. PMID: 21916437
- Omuralide and vibralactone: differences in the proteasome- β-lactone-γ-lactam binding scaffold alter target preferences. | List, A., et al. 2014. Angew Chem Int Ed Engl. 53: 571-4. PMID: 24285701
- Stereocontrolled access to δ-lactone-fused-γ-lactams bearing angular benzylic quaternary stereocenters. | Beng, TK., et al. 2022. RSC Adv. 12: 17617-17620. PMID: 35765420
- Inhibition of proteasome activities and subunit-specific amino-terminal threonine modification by lactacystin. | Fenteany, G., et al. 1995. Science. 268: 726-31. PMID: 7732382
- Mechanistic studies on the inactivation of the proteasome by lactacystin in cultured cells. | Dick, LR., et al. 1997. J Biol Chem. 272: 182-8. PMID: 8995245
- Total synthesis and biological activity of lactacystin, omuralide and analogs. | Corey, EJ. and Li, WD. 1999. Chem Pharm Bull (Tokyo). 47: 1-10. PMID: 9987821
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
clasto-Lactacystin β-lactone, 100 µg | sc-202106 | 100 µg | $254.00 |
