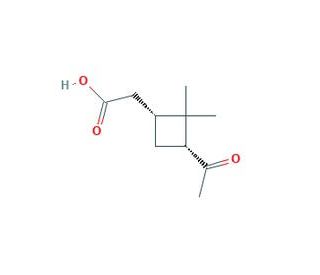
cis-Pinonic acid (CAS 61826-55-9)
QUICK LINKS
cis-Pinonic acid is a compound that arises from the oxidation of pinene, a major component of pine resin. This organic acid features a bicyclic structure with a ketone functional group, which plays a key role in its chemical reactivity. It is involved in studies that explore atmospheric chemistry, particularly the processes that affect organic aerosol formation and behavior. cis-Pinonic acid acts as an intermediate in the synthesis of more complex organic molecules, often serving as a precursor in the formation of secondary organic aerosols. Its role in these transformations is for understanding the lifecycle of organic compounds in the atmosphere and their impact on air quality and climate. The compound′s ability to participate in various chemical reactions makes it a subject of interest in environmental chemistry, where it helps reveal the pathways of biogenic volatile organic compounds in the atmosphere.
cis-Pinonic acid (CAS 61826-55-9) References
- Evaporation rates and vapor pressures of individual aerosol species formed in the atmospheric oxidation of alpha- and beta-pinene. | Bilde, M. and Pandis, SN. 2001. Environ Sci Technol. 35: 3344-9. PMID: 11529575
- Nonderivatization analytical method of fatty acids and cis pinonic acid and its application in ambient PM2.5 aerosols in the greater Vancouver area in Canada. | Cheng, Y. and Li, SM. 2005. Environ Sci Technol. 39: 2239-46. PMID: 15871259
- Roadside, urban, and rural comparison of primary and secondary organic molecular markers in ambient PM2.5. | Yan, B., et al. 2009. Environ Sci Technol. 43: 4287-93. PMID: 19603636
- Experimental and theoretical study of aqueous cis-pinonic acid photolysis. | Lignell, H., et al. 2013. J Phys Chem A. 117: 12930-45. PMID: 24245507
- Heterogeneous Kinetics of cis-Pinonic Acid with Hydroxyl Radical under Different Environmental Conditions. | Lai, C., et al. 2015. J Phys Chem A. 119: 6583-93. PMID: 26017096
- OH-Radical Oxidation of Surface-Active cis-Pinonic Acid at the Air-Water Interface. | Enami, S. and Sakamoto, Y. 2016. J Phys Chem A. 120: 3578-87. PMID: 27098046
- Structures and energetics of hydrated deprotonated cis-pinonic acid anion clusters and their atmospheric relevance. | Hou, GL., et al. 2017. Phys Chem Chem Phys. 19: 10676-10684. PMID: 28398433
- Efficient scavenging of Criegee intermediates on water by surface-active cis-pinonic acid. | Enami, S. and Colussi, AJ. 2017. Phys Chem Chem Phys. 19: 17044-17051. PMID: 28643829
- cis-Pinonic Acid Oxidation by Hydroxyl Radicals in the Aqueous Phase under Acidic and Basic Conditions: Kinetics and Mechanism. | Witkowski, B. and Gierczak, T. 2017. Environ Sci Technol. 51: 9765-9773. PMID: 28719200
- Reactions of Criegee Intermediates with Benzoic Acid at the Gas/Liquid Interface. | Qiu, J., et al. 2018. J Phys Chem A. 122: 6303-6310. PMID: 29989413
- Theoretical study of the cis-pinonic acid and its atmospheric hydrolysate participation in the atmospheric nucleation. | Shi, X., et al. 2019. Sci Total Environ. 674: 234-241. PMID: 31005830
- Physicochemical Properties of Pinic, Pinonic, Norpinic, and Norpinonic Acids as Relevant α-Pinene Oxidation Products. | Kołodziejczyk, A., et al. 2019. J Phys Chem B. 123: 8261-8267. PMID: 31455074
- Synthesis of Carboxylic Acid and Dimer Ester Surrogates to Constrain the Abundance and Distribution of Molecular Products in α-Pinene and β-Pinene Secondary Organic Aerosol. | Kenseth, CM., et al. 2020. Environ Sci Technol. 54: 12829-12839. PMID: 32813970
- pH Dependence of the OH Reactivity of Organic Acids in the Aqueous Phase. | Amorim, JV., et al. 2020. Environ Sci Technol. 54: 12484-12492. PMID: 32936620
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
cis-Pinonic acid, 5 g | sc-227677 | 5 g | $93.00 |
