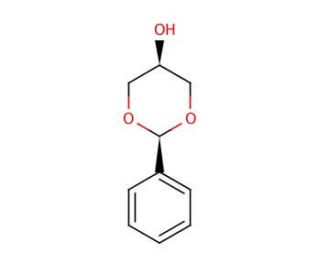
cis-1,3-O-Benzylideneglycerol (CAS 4141-19-9)
QUICK LINKS
cis-1,3-O-Benzylideneglycerol, listed under the CAS number 4141-19-9, is a synthetic organic compound characterized by a glycerol backbone in which the hydroxyl groups at the 1 and 3 positions are protected by a benzylidene acetal linkage. This structural modification renders the central hydroxyl group at the 2 position more reactive, making it a focal point for further chemical modifications. In research, cis-1,3-O-Benzylideneglycerol is widely used as an intermediate in the synthesis of more complex glycerol derivatives. This compound is particularly valuable in the study of lipid chemistry and in the synthesis of biologically relevant molecules where the protected form of glycerol can undergo selective reactions without interference from the other hydroxyl groups. By employing this compound, chemists can explore various synthetic pathways, including esterification and etherification reactions, which are crucial for developing advanced materials and fine chemicals. Additionally, its use extends to the field of polymer chemistry, where it acts as a building block for creating novel polymers with specific properties influenced by the glycerol moiety. The benzylidene group in cis-1,3-O-Benzylideneglycerol offers steric hindrance, which is exploited in stereoselective synthesis, providing insights into reaction mechanisms and the development of new synthetic methodologies. Through these applications, this chemical plays a significant role in advancing research in organic synthesis and material science.
cis-1,3-O-Benzylideneglycerol (CAS 4141-19-9) References
- Syntheses of 1-[(2-hydroxyethoxy)methyl]- and 1-[(1,3-dihydroxy-2-propoxy)methyl]- derivatives of 5-substituted-2,4-difluorobenzene: unnatural acyclo thymidine mimics for evaluation as anticancer and antiviral agents. | Wang, ZX., et al. 2000. Nucleosides Nucleotides Nucleic Acids. 19: 1397-411. PMID: 11092311
- Synthesis and characterization of polyether-ester dendrimers from glycerol and lactic acid. | Carnahan, MA. and Grinstaff, MW. 2001. J Am Chem Soc. 123: 2905-6. PMID: 11456987
- A short, concise route to diphosphatidylglycerol (cardiolipin) and its variants. | Krishna, UM., et al. 2004. Lipids. 39: 595-600. PMID: 15554161
- Evaluation of the immediate vascular stability of lipoprotein lipase-generated 2-monoacylglycerol in mice. | Kleberg, K., et al. 2014. Biofactors. 40: 596-602. PMID: 25359532
- Targeted metabolomics shows plasticity in the evolution of signaling lipids and uncovers old and new endocannabinoids in the plant kingdom. | Gachet, MS., et al. 2017. Sci Rep. 7: 41177. PMID: 28120902
- Inhibition of Tetrahydrofuran Hydrate Formation in the Presence of Polyol-Modified Glass Surfaces. | Hall, JR. and Baures, PW. 2017. Energy Fuels. 31: 7816-7823. PMID: 35444363
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
cis-1,3-O-Benzylideneglycerol, 10 g | sc-211107 | 10 g | $360.00 |
