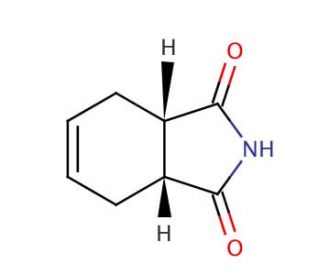
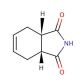
cis-1,2,3,6-Tetrahydrophthalimide (CAS 1469-48-3)
QUICK LINKS
Cis-1,2,3,6-Tetrahydrophthalimide (THPI) is a cyclic organic compound belonging to the phthalimide family, characterized by four interconnected rings of carbon and nitrogen atoms. This versatile compound has found widespread use in diverse scientific research applications. One of its significant roles lies in acting as a ligand for the synthesis of metal complexes, a catalyst for polymer synthesis, and a reagent for various organic compound syntheses. Moreover, it has been extensively employed in biochemical studies, encompassing investigations into enzyme kinetics and cell signaling pathways. Additionally, in laboratory experiments, cis-1,2,3,6-Tetrahydrophthalimide has proven valuable for studying the effects of different compounds on cellular processes. Although the precise mechanism of action of cis-1,2,3,6-Tetrahydrophthalimide remains somewhat elusive, it is thought to serve as an inhibitor of drug-metabolizing enzymes and the enzyme cyclooxygenase-2 (COX-2). Furthermore, research has shown that this compound binds to specific proteins within cell membranes, suggesting potential involvement in regulating cellular processes. The compound cis-1,2,3,6-Tetrahydrophthalimide holds significant importance in the scientific community, offering a wide array of applications and promising insights into various biochemical phenomena, though further investigations are required to fully comprehend its intricate mechanisms of action.
cis-1,2,3,6-Tetrahydrophthalimide (CAS 1469-48-3) References
- WITHDRAWN: Derivatives of cis-1,2,3,6-tetrahydrophthalimide for the treatment of benign prostatic hyperplasia. | Sarma, PK., et al. 2005. Bioorg Med Chem Lett.. PMID: 16275082
- Captan exposure and evaluation of a pesticide exposure algorithm among orchard pesticide applicators in the Agricultural Health Study. | Hines, CJ., et al. 2008. Ann Occup Hyg. 52: 153-66. PMID: 18326518
- Synthesis, anti-inflammatory and anticancer activity evaluation of some novel acridine derivatives. | Sondhi, SM., et al. 2010. Eur J Med Chem. 45: 555-63. PMID: 19926172
- Liquid chromatography-tandem mass spectrometry (LC/APCI-MS/MS) methods for the quantification of captan and folpet phthalimide metabolites in human plasma and urine. | Berthet, A., et al. 2011. Anal Bioanal Chem. 399: 2243-55. PMID: 21229238
- Imidase catalyzing desymmetric imide hydrolysis forming optically active 3-substituted glutaric acid monoamides for the synthesis of gamma-aminobutyric acid (GABA) analogs. | Nojiri, M., et al. 2015. Appl Microbiol Biotechnol. 99: 9961-9. PMID: 26205522
- Urinary biomarker concentrations of captan, chlormequat, chlorpyrifos and cypermethrin in UK adults and children living near agricultural land. | Galea, KS., et al. 2015. J Expo Sci Environ Epidemiol. 25: 623-31. PMID: 26374656
- Overcoming difficulties in the evaluation of captan and folpet residues by supercritical fluid chromatography coupled to mass spectrometry. | Cutillas, V., et al. 2021. Talanta. 223: 121714. PMID: 33303162
- Assessment of Environmental Pollution and Human Exposure to Pesticides by Wastewater Analysis in a Seven-Year Study in Athens, Greece. | Rousis, NI., et al. 2021. Toxics. 9: PMID: 34678955
- Mitosis Inhibitors Induce Massive Accumulation of Phytoene in the Microalga Dunaliella salina. | Xu, Y. and Harvey, PJ. 2021. Mar Drugs. 19: PMID: 34822466
- Evaluation of the phycoremediation potential of microalgae for captan removal: Comprehensive analysis on toxicity, detoxification and antioxidants modulation. | Hamed, SM., et al. 2022. J Hazard Mater. 427: 128177. PMID: 34999404
- Enzymatic resolution of N-Hydroxymethyl γ-Butyrolactams. An access to optically active γ-Butyrolactams | Bruno Jouglet, Gérard Rousseau ∗. 1993. Tetrahedron Letters. 34: 2307-2310.
- Formation of polychlorinated dibenzo-p-dioxins and dibenzofurans (PCDD/F) in oxidation of captan pesticide | Kai Chen a, Dominika Wojtalewicz a, Mohammednoor Altarawneh b, John C. Mackie a c, Eric M. Kennedy a, Bogdan Z. Dlugogorski a. 2011. Proceedings of the Combustion Institute. 33: 701-708.
- Efficient Catalytic Hydrogenation of N-Unsubstituted Cyclic Imides to Cyclic Amines | Dr. Anna M. Maj, Dr. Isabelle Suisse, Dr. Nathalie Pinault, Dr. Nicolas Robert, Dr. Francine Agbossou-Niedercorn. 2014. 6: 2621-2625.
- Quantitation of phenyl-amide and phthalimide fungicide formulations in solid-state via UV–MALDI mass spectrometry-matrix effects in soils | Bojidarka Ivanova & Michael Spiteller. 2015. Journal of Soils and Sediments. 15: 917–925.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
cis-1,2,3,6-Tetrahydrophthalimide, 100 g | sc-257248 | 100 g | $32.00 |
