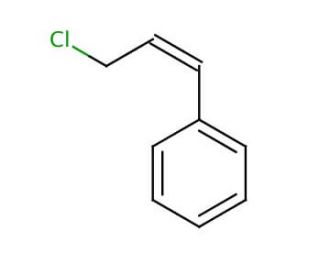
Molecular structure of Cinnamyl chloride, CAS Number: 2687-12-9
Cinnamyl chloride (CAS 2687-12-9)
Alternate Names:
(3-Chloropropenyl)benzene
CAS Number:
2687-12-9
Molecular Weight:
152.62
Molecular Formula:
C9H9Cl
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Cinnamyl chloride is a chemical compound that functions as a reactive intermediate in organic synthesis. It acts as an important building block in the production of various compounds, including agrochemicals and fragrances. Its mechanism of action involves participating in nucleophilic substitution reactions, where the chlorine atom is replaced by a nucleophile to form new carbon-carbon or carbon-heteroatom bonds. Cinnamyl Chloride′s reactivity allows for the introduction of the cinnamyl group into different molecular frameworks, enabling the synthesis of diverse organic molecules.
Cinnamyl chloride (CAS 2687-12-9) References
- [Syntheses and antitumor activities of the derivatives of P-methyl goniotriol in vitro]. | Chen, H., et al. 2002. Yao Xue Xue Bao. 37: 775-80. PMID: 12567860
- Origin of the regio- and stereoselectivity in palladium-catalyzed electrophilic substitution via bis(allyl)palladium complexes. | Wallner, OA. and Szabó, KJ. 2003. Chemistry. 9: 4025-30. PMID: 12953188
- First enantioselective allylic etherification with phenols catalyzed by chiral ruthenium bisoxazoline complexes. | Mbaye, MD., et al. 2004. Chem Commun (Camb). 1870-1. PMID: 15306925
- Primary sensitization to cinnamyl chloride in an operator of a pharmaceutical company. | Goossens, A., et al. 2006. Contact Dermatitis. 55: 364-5. PMID: 17101013
- Metal-mediated allylation of enzymatically oxidized methyl α-D-galactopyranoside. | Leppänen, AS., et al. 2010. Carbohydr Res. 345: 2610-5. PMID: 21055730
- On the Importance of the Aromatic Ring Parameter in Studies of the Solvolyses of Cinnamyl and Cinnamoyl Halides. | D'Souza, MJ., et al. 2010. Org Chem Int. 2010: 1-9. PMID: 21552456
- SCS-pincer palladium-catalyzed auto-tandem catalysis using dendritic catalysts in semi-permeable compartments. | Pijnenburg, NJ., et al. 2011. Dalton Trans. 40: 8896-905. PMID: 21731937
- Mechanistic studies on the SCS-pincer palladium(II)-catalyzed tandem stannylation/electrophilic allylic substitution of allyl chlorides with hexamethylditin and benzaldehydes. | Pijnenburg, NJ., et al. 2013. Chemistry. 19: 4858-68. PMID: 23436343
- Heterogeneous rate constants for indium mediated allylations: cinnamyl chloride in ethanol/water mixtures. | Hill, AN., et al. 2013. J Phys Chem A. 117: 8826-35. PMID: 23879332
- Silver encapsulated copper salen complex: efficient catalyst for electrocarboxylation of cinnamyl chloride with CO2. | Wu, LX., et al. 2019. RSC Adv. 9: 32628-32633. PMID: 35529726
- A sensitive LC-MS/MS method for the determination of potential genotoxic impurities in Cinnarizine. | Mullangi, S., et al. 2023. Ann Pharm Fr. 81: 74-82. PMID: 35792147
- Direct C-H Allylation of Unactivated Alkanes by Cooperative W/Cu Photocatalysis. | Martínez-Balart, P., et al. 2022. Org Lett. 24: 6874-6879. PMID: 36098628
- Chiral aldehyde catalysis enables direct asymmetric α-substitution reaction of N-unprotected amino acids with halohydrocarbons. | Shen, HR., et al. 2023. Chem Sci. 14: 5665-5671. PMID: 37265737
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Cinnamyl chloride, 5 g | sc-234366 | 5 g | $37.00 |
