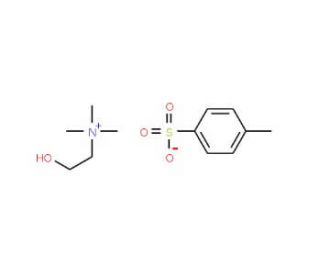
Choline p-toluenesulfonate salt (CAS 55357-38-5)
QUICK LINKS
Choline p-toluenesulfonate salt is a chemical compound that has been instrumental in various fields of research, particularly in organic chemistry and materials science. It consists of choline, a quaternary ammonium salt, paired with p-toluenesulfonic acid, an organosulfur compound. This configuration provides an ionic structure that is particularly useful for studying ionic interactions and solvent dynamics in non-aqueous environments. Choline p-toluenesulfonate salt is utilized extensively to investigate the phase behavior of ionic liquids and their applications as solvents in organic synthesis and electrochemical processes. In research settings, its ability to form stable, conductive solutions is exploited to enhance the understanding of charge transport mechanisms in ionic systems. Additionally, this compound aids in the study of catalysis processes, where its strong ionic nature can influence the reactivity and selectivity of catalytic reactions. The properties of choline p-toluenesulfonate salt make it a valuable tool in the development of novel catalytic systems and in the enhancement of green chemistry protocols, focusing on reducing the environmental impact of chemical processes through more efficient and selective reactions. This research has significant implications for advancing fundamental science in the manipulation and application of ionic compounds in research.
Choline p-toluenesulfonate salt (CAS 55357-38-5) References
- Rapid synthesis of 2-desoxy-2-amino-3-phosphocholine-glycerinic-acid- alkylester, 1-alkyl-1-desoxy- and 1-O-alkyl-2-desoxy-2-amino-sn-glycero-3- phosphocholines, -3-phospho-N,N'-dimethylethanolamine and -3-phospho-Fmoc- serine-methylester. | Deigner, HP. and Fyrnys, B. 1992. Chem Phys Lipids. 61: 199-208. PMID: 1511493
- Synthesis of oxidized phospholipids by sn-1 acyltransferase using 2-15-HETE lysophospholipids. | Liu, GY., et al. 2019. J Biol Chem. 294: 10146-10159. PMID: 31080170
- Structural Determination of Lysosphingomyelin-509 and Discovery of Novel Class Lipids from Patients with Niemann-Pick Disease Type C. | Maekawa, M., et al. 2019. Int J Mol Sci. 20: PMID: 31658747
- An effective reagent to functionalize alcohols with phosphocholine. | Xu, LL., et al. 2020. Org Biomol Chem. 18: 767-770. PMID: 31912847
- Zwitterionic Phosphodiester-Substituted Neoglycoconjugates as Ligands for Antibodies and Acute Phase Proteins. | Labrada, KP., et al. 2020. ACS Chem Biol. 15: 369-377. PMID: 31935056
- Synthesis of ether lipids: natural compounds and analogues. | Gomes, MAGB., et al. 2023. Beilstein J Org Chem. 19: 1299-1369. PMID: 37701305
- Synthesis of phosphatidylcholine analogs with an alkyl group at C1 or C3 of the glycerol moiety. | Witzke, NM. and Bittman, R. 1985. J Lipid Res. 26: 623-8. PMID: 4020301
- Analogs of natural lipids. VII. Synthesis of cyclopentanoid analogs of phosphatidylcholine. | Hancock, AJ., et al. 1982. J Lipid Res. 23: 183-9. PMID: 7057105
- A lipid-lipase aggregate with ether linkage as a new type of immobilized enzyme for enantioselective hydrolysis in organic solvents | AKITA, H., UMEZAWA, I., MATSUKURA, H., & OISHI, T. 1992. Chemical and pharmaceutical bulletin. 40(2): 318-324.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Choline p-toluenesulfonate salt, 5 g | sc-281523 | 5 g | $416.00 | |||
Choline p-toluenesulfonate salt, 10 g | sc-281523A | 10 g | $624.00 |
