Chlorotriethoxysilane (CAS 4667-99-6)
Available in US only.
QUICK LINKS
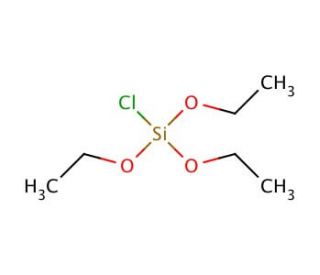
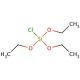
Chlorotriethoxysilane (CTES) is a transparent liquid compound characterized by the chemical formula Cl(OC2H5)3Si. It is an active organosilicon compound widely employed in numerous applications, including catalysis, surfactant usage, reagent functionality, and as a precursor in synthesizing diverse silanes. With its versatility, Chlorotriethoxysilane finds utility in laboratory applications, industrial processes, and even household applications. Chlorotriethoxysilane serves as a fundamental reagent in organic synthesis. It facilitates the conversion of alcohols into alkyl chlorides and participates in the formation of ethers, esters, and various functional groups. Moreover, it plays a role in the synthesis of silanes, which encompass silicon-oxygen bonds. Additionally, Chlorotriethoxysilane contributes to the production of polysiloxanes, which represent polymers comprising recurring siloxane units. Due to its highly reactive nature, Chlorotriethoxysilane readily engages in chemical reactions with other compounds. It reacts with alcohols to generate alkyl chlorides and with amines to produce amides. Furthermore, it can form esters when reacting with acids. Notably, the hydrolysis of the silanol group through the reaction of Chlorotriethoxysilane with water holds particular significance as a pivotal step in the synthesis of silanes and polysiloxanes.
Chlorotriethoxysilane (CAS 4667-99-6) References
- Synthesis and properties of 1,3,5-benzene periodic mesoporous organosilica (PMO): novel aromatic PMO with three point attachments and unique thermal transformations. | Kuroki, M., et al. 2002. J Am Chem Soc. 124: 13886-95. PMID: 12431120
- Stable silanetriols that contain tert-alkoxy groups: versatile precursors of siloxane-based nanomaterials. | Suzuki, J., et al. 2008. Chemistry. 14: 973-80. PMID: 17992681
- Modification of micro-cantilever sensors with sol-gels to enhance performance and immobilize chemically selective phases. | Fagan, BC., et al. 2000. Talanta. 53: 599-608. PMID: 18968147
- Synthesis of 4-aryl- and 4-alkyl-2-silyl-1,3-butadienes and their Diels-Alder/cross-coupling reactions. | Junker, CS., et al. 2010. J Org Chem. 75: 8155-65. PMID: 21069964
- Dendritic, nanosized building block for siloxane-based materials: a spherosilicate dendrimer. | Kawahara, K., et al. 2011. Chemistry. 17: 13188-96. PMID: 22038556
- Functionalized mesoporous silica thin films as a tunable nonlinear optical material. | Laskowska, M., et al. 2017. Nanoscale. 9: 12110-12123. PMID: 28800139
- Photovoltaic Properties and Long-Term Durability of Porphyrin-Sensitized Solar Cells with Silicon-Based Anchoring Groups. | Higashino, T., et al. 2017. ACS Omega. 2: 6958-6967. PMID: 31457279
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Chlorotriethoxysilane, 25 g | sc-234347 | 25 g | $203.00 | |||
| US: Only available in the US | ||||||
