Chlorothricin (CAS 34707-92-1)
See product citations (1)
QUICK LINKS
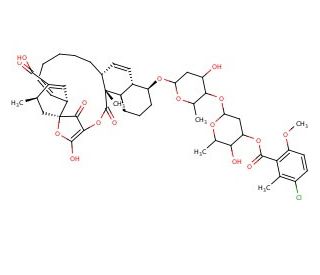
Chlorothricin, with the CAS number 34707-92-1, is an antibiotic compound produced by the bacterium Streptomyces antibioticus. It is a complex molecule that belongs to the spiroketal class of polyketides, known for their complex and diverse structures. Chlorothricin features a unique combination of spiroketal and macrolactone rings, which are key to its biological activity. The primary mechanism of action of Chlorothricin involves the inhibition of bacterial DNA gyrase, an enzyme crucial for DNA replication and transcription. By binding to and inhibiting this enzyme, Chlorothricin disrupts the supercoiling process essential for the proper functioning of bacterial DNA, leading to an effective halt in bacterial cell replication and growth. In research, Chlorothricin has been extensively used as a tool to study the enzymatic processes related to DNA supercoiling and to understand bacterial resistance mechanisms. Its role in inhibiting DNA gyrase, a target for antibacterial strategies, makes it valuable for probing the efficacy and mechanism of action of similar compounds and for investigating potential pathways for overcoming bacterial resistance. Moreover, the structural complexity of Chlorothricin provides a rich area for chemical synthesis studies, aiming to explore the relationships between molecular structure and bioactivity.
Chlorothricin (CAS 34707-92-1) References
- Mode of action of the macrolide-type antibiotic, chlorothricin. Effect of the antibiotic on the catalytic activity and some structural parameters of pyruvate carboxylases purified from rat and chicken liver. | Schindler, PW. and Scrutton, MC. 1975. Eur J Biochem. 55: 543-53. PMID: 1175611
- New cholesterol biosynthesis inhibitors MC-031 (O-demethylchlorothricin), -032 (O-demethylhydroxychlorothricin), -033 and -034. | Kawashima, A., et al. 1992. J Antibiot (Tokyo). 45: 207-12. PMID: 1556012
- Genetic characterization of the chlorothricin gene cluster as a model for spirotetronate antibiotic biosynthesis. | Jia, XY., et al. 2006. Chem Biol. 13: 575-85. PMID: 16793515
- Chlorothricin, and inhibitor of porcine-heart malate dehydrogenases, discriminating between the mitochondrial and cytoplasmic isoenzyme. | Schindler, PW. 1975. Eur J Biochem. 51: 579-85. PMID: 168072
- Apoptosis and inactivation of the PI3-kinase pathway by tetrocarcin A in breast cancers. | Nakajima, H., et al. 2007. Biochem Biophys Res Commun. 356: 260-5. PMID: 17350598
- Glyceryl-S-acyl carrier protein as an intermediate in the biosynthesis of tetronate antibiotics. | Sun, Y., et al. 2008. Chembiochem. 9: 150-6. PMID: 18046685
- Dissection of two acyl-transfer reactions centered on acyl-S-carrier protein intermediates for incorporating 5-chloro-6-methyl-O-methylsalicyclic acid into chlorothricin. | He, QL., et al. 2009. Chembiochem. 10: 813-9. PMID: 19266533
- Insights into bacterial 6-methylsalicylic acid synthase and its engineering to orsellinic acid synthase for spirotetronate generation. | Ding, W., et al. 2010. Chem Biol. 17: 495-503. PMID: 20534347
- Coordinative Modulation of Chlorothricin Biosynthesis by Binding of the Glycosylated Intermediates and End Product to a Responsive Regulator ChlF1. | Li, Y., et al. 2016. J Biol Chem. 291: 5406-17. PMID: 26750095
- Co-expression of a SARP Family Activator ChlF2 and a Type II Thioesterase ChlK Led to High Production of Chlorothricin in Streptomyces antibioticus DSM 40725. | Li, Y., et al. 2020. Front Bioeng Biotechnol. 8: 1013. PMID: 32974326
- Structural Insight of KSIII (β-Ketoacyl-ACP Synthase)-like Acyltransferase ChlB3 in the Biosynthesis of Chlorothricin. | Saeed, AU., et al. 2022. Molecules. 27: PMID: 36234941
- Mode of action of the macrolide-type antibiotic, chlorothricin. Kinetic study of the inhibition of pyruvate carboxylase from Bacillus stearothermophilus. | Schindler, PW. and Zähner, H. 1973. Eur J Biochem. 39: 591-600. PMID: 4775066
- Biosynthesis of the macrolide antibiotic chlorothricin. | Mascaretti, OA., et al. 1981. Biochemistry. 20: 919-24. PMID: 7011379
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Chlorothricin, 1 mg | sc-202098 | 1 mg | $183.00 |
