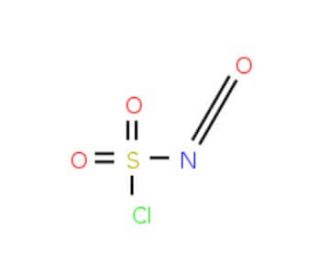
Chlorosulfonyl isocyanate (CAS 1189-71-5)
QUICK LINKS
Chlorosulfonyl isocyanate (CSI), a highly reactive and versatile compound, serves as a significant reagent in organic synthesis. This colorless to light yellow liquid possesses a pungent odor. With its powerful electrophilic nature, Chlorosulfonyl isocyanate finds application in various reactions, enabling the synthesis of a diverse array of organic compounds. It proves valuable in the synthesis of heterocycles, polymers, and other organic compounds. The widespread use of chlorosulfonyl isocyanate lies in its role as a reagent for organic synthesis. The reactivity of chlorosulfonyl isocyanate stems from its ability to act as an electrophile. This property allows it to readily react with nucleophiles such as amines, alcohols, and acids, facilitating the formation of diverse organic compounds.
Chlorosulfonyl isocyanate (CAS 1189-71-5) References
- Photofragmentation Mechanisms of Chlorosulfonyl Isocyanate, ClSO2NCO, Excited with Synchrotron Radiation between 12 and 550 eV. | Moreno Betancourt, A., et al. 2015. J Phys Chem A. 119: 8021-30. PMID: 26121558
- The synthesis of novel sulfamides derived from β-benzylphenethylamines as acetylcholinesterase, butyrylcholinesterase and carbonic anhydrase enzymes inhibitors. | Akıncıoğlu, A., et al. 2017. Bioorg Chem. 74: 238-250. PMID: 28866249
- Radiosynthesis of a carbon-11-labeled AMPAR allosteric modulator as a new PET radioligand candidate for imaging of Alzheimer's disease. | Miao, C., et al. 2019. Bioorg Med Chem Lett. 29: 1177-1181. PMID: 30922660
- Synthesis of β-amino acid derivatives and their inhibitory profiles against some metabolic enzymes. | Atmaca, U., et al. 2019. Arch Pharm (Weinheim). 352: e1900200. PMID: 31544276
- Uracil-5-yl O-Sulfamate: An Illusive Radiosensitizer. Pitfalls in Modeling the Radiosensitizing Derivatives of Nucleobases. | Spisz, P., et al. 2020. J Phys Chem B. 124: 5600-5613. PMID: 32539395
- Development of sulfahydantoin derivatives as β-lactamase inhibitors. | Paquet-Côté, PA., et al. 2021. Bioorg Med Chem Lett. 35: 127781. PMID: 33422604
- Evolution of the Synthesis of Remdesivir. Classical Approaches and Most Recent Advances. | Vargas, DF., et al. 2021. ACS Omega. 6: 19356-19363. PMID: 34368522
- Non-Genetic Generation of Antibody Conjugates Based on Chemoenzymatic Tyrosine Click Chemistry. | Bruins, JJ., et al. 2021. Bioconjug Chem. 32: 2167-2172. PMID: 34519477
- The (±)-6-Aza[1.0]triblattane Skeleton: Contraction beyond the Wilder-Culberson Ring System. | Fahrenhorst-Jones, T., et al. 2022. Org Lett. 24: 903-906. PMID: 35043631
- A safe alternative synthesis of primary carbamates from alcohols; in vitro and in silico assessments as an alternative acetylcholinesterase inhibitors. | Atmaca, U., et al. 2022. J Biomol Struct Dyn. 1-10. PMID: 36224670
- 1,2,3-Benzoxathiazine-2,2-dioxides - effective inhibitors of human carbonic anhydrases. | Ivanova, J., et al. 2023. J Enzyme Inhib Med Chem. 38: 225-238. PMID: 36373195
- Novel pseudonucleosides and sulfamoyl-oxazolidinone β-D-glucosamine derivative as anti-COVID-19: design, synthesis, and in silico study. | Mansouri, R., et al. 2023. J Biomol Struct Dyn. 1-18. PMID: 37098814
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Chlorosulfonyl isocyanate, 25 g | sc-255017 | 25 g | $38.00 | |||
Chlorosulfonyl isocyanate, 100 g | sc-255017A | 100 g | $142.00 |
