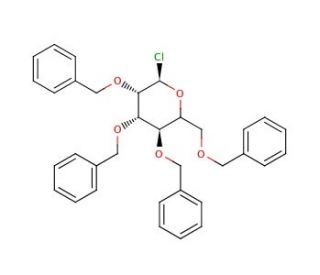
Chloro 2,3,4,6-Tetra-O-benzyl-α-D-glucopyranoside (CAS 25320-59-6)
QUICK LINKS
Chloro 2,3,4,6-Tetra-O-benzyl-α-D-glucopyranoside, a derivative of glucose, has garnered significant interest in scientific research due to its versatility and potential applications across various fields. As a glycosylation reagent, it serves as a key intermediate in the synthesis of complex carbohydrates and glycoconjugates, playing a crucial role in the study of glycosylation reactions and the development of novel carbohydrate-based materials. Its mechanism of action involves the protection of the hydroxyl groups of glucose with benzyl groups, allowing for selective manipulation of specific hydroxyl groups during subsequent glycosylation reactions. This chemical has been extensively utilized in organic synthesis and carbohydrate chemistry, enabling the synthesis of diverse glycosidic linkages and glycoconjugates with precise control over stereochemistry and regiochemistry. Moreover, Chloro 2,3,4,6-Tetra-O-benzyl-α-D-glucopyranoside has found applications in the synthesis of glycopeptides, glycolipids, and glycoproteins, facilitating the study of carbohydrate-protein interactions, cellular recognition processes, and the development of carbohydrate-based vaccines. Furthermore, its compatibility with various protecting groups and glycosylation methodologies makes it a valuable tool in the synthesis of complex oligosaccharides and glycoconjugates for biological and pharmaceutical research purposes. Overall, Chloro 2,3,4,6-Tetra-O-benzyl-α-D-glucopyranoside stands as a fundamental building block in carbohydrate chemistry, enabling the synthesis of structurally diverse and biologically relevant glycoconjugates for research applications.
Chloro 2,3,4,6-Tetra-O-benzyl-α-D-glucopyranoside (CAS 25320-59-6) References
- Bausteine von Oligosacchariden, LXXIV. Synthese von verzweigten Tri‐und Tetrasaccharidsequenzen der „Repeating Unit" ︁ der O‐spezifischen Kette des Lipopolysaccharides aus Aeromonas salmonicida | Paulsen, H., & Lorentzen, J. P. 1986. Liebigs Annalen der Chemie. 9: 1586-1599.
- Bausteine von Oligosacchariden, LXXXIII. Synthese der Repeating Unit der Teichuronsäure von Micrococcus luteus | Paulsen, H., Helpap, B., & Lorentzen, J. P. 1987. Liebigs Annalen der Chemie. 5: 431-437.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Chloro 2,3,4,6-Tetra-O-benzyl-α-D-glucopyranoside, 1 g | sc-221425 | 1 g | $490.00 |
