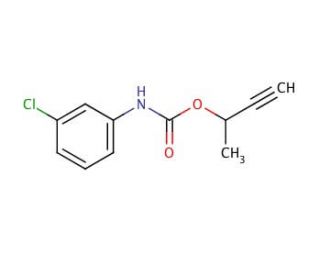
Chlorbufam (CAS 1967-16-4)
QUICK LINKS
Chlorbufam, a synthetic herbicide, finds application in agriculture and horticulture for weed control, targeting both grasses and broadleaves. Belonging to the sulfonylurea herbicide class, it has been utilized since the late 1960s. Operating as a non-selective herbicide, Chlorbufam exhibits efficacy against a wide spectrum of weed species, without distinguishing between weeds and other plants. However, caution must be exercised as it poses toxicity risks to humans, animals, and the environment if not appropriately employed. Extensive scientific research has focused on Chlorbufam, exploring various applications. It has been instrumental in investigating the impact of herbicides on plant growth, development, and soil microbial communities. Additionally, Chlorbufam has facilitated studies on herbicide effects on aquatic ecosystems and human health. Its mode of action involves the inhibition of acetolactate synthase (ALS), an enzyme for the biosynthesis of the amino acids valine and isoleucine. By hindering ALS, Chlorbufam disrupts the production of these vital amino acids, leading to growth inhibition in the plant. This mechanism aligns with other sulfonylurea herbicides like metsulfuron-methyl and flazasulfuron.
Chlorbufam (CAS 1967-16-4) References
- Multiresidue method for the determination of residues of 251 pesticides in fruits and vegetables by gas chromatography/mass spectrometry and liquid chromatography with fluorescence detection. | Fillion, J., et al. 2000. J AOAC Int. 83: 698-713. PMID: 10868594
- Herbicide and plant growth regulator analysis by capillary electrophoresis. | Eash, DT. and Bushway, RJ. 2000. J Chromatogr A. 880: 281-94. PMID: 10890524
- TOPS-MODE based QSARs derived from heterogeneous series of compounds. Applications to the design of new herbicides. | Pérez González, M., et al. 2003. J Chem Inf Comput Sci. 43: 1192-9. PMID: 12870911
- Determination of 266 pesticide residues in apple juice by matrix solid-phase dispersion and gas chromatography-mass selective detection. | Chu, XG., et al. 2005. J Chromatogr A. 1063: 201-10. PMID: 15700472
- Surfactant effect on persistence of oxadiazolyl 3(2H)-pyridazinones against Pseudaletia separata walker. | Huang, Q., et al. 2007. J Environ Sci Health B. 42: 305-10. PMID: 17454384
- Surface swabbing technique for the rapid screening for pesticides using ambient pressure desorption ionization with high-resolution mass spectrometry. | Edison, SE., et al. 2011. Rapid Commun Mass Spectrom. 25: 127-39. PMID: 21157860
- A Quantitative Structure Activity Relationship for acute oral toxicity of pesticides on rats: Validation, domain of application and prediction. | Hamadache, M., et al. 2016. J Hazard Mater. 303: 28-40. PMID: 26513561
- Multiclass method for pesticides quantification in honey by means of modified QuEChERS and UHPLC-MS/MS. | Tette, PA., et al. 2016. Food Chem. 211: 130-9. PMID: 27283616
- Agricultural exposures to carbamate herbicides and fungicides and central nervous system tumour incidence in the cohort AGRICAN. | Piel, C., et al. 2019. Environ Int. 130: 104876. PMID: 31344646
- Qualitative screening and quantitative determination of 569 pesticide residues in honeysuckle using ultrahigh-performance liquid chromatography coupled to quadrupole-Orbitrap high resolution mass spectrometry. | Zhou, H., et al. 2019. J Chromatogr A. 1606: 460374. PMID: 31447205
- Comparison of the extraction efficiency of ice concentration linked with extractive stirrer, stir bar sorptive extraction, and solid-phase microextraction for pesticides from drinking water. | Skaggs, CS., et al. 2020. J Chromatogr A. 1622: 461102. PMID: 32389418
- Method validation, residue and risk assessment of 260 pesticides in some leafy vegetables using liquid chromatography coupled to tandem mass spectrometry. | Balkan, T. and Yılmaz, Ö. 2022. Food Chem. 384: 132516. PMID: 35240574
- Automated determination of phenylcarbamate herbicides in environmental waters by on-line trace enrichment and reversed-phase liquid chromatography-diode array detection. | Hidalgo, C., et al. 1998. J Chromatogr A. 823: 121-8. PMID: 9818399
- Phototransformation of three herbicides: chlorbufam, isoproturon, and chlorotoluron. Influence of irradiation on toxicity | C. Tixier,1L. Meunier,3F. Bonnemoy,2and P. Boule3. 2000. International Journal of Photoenergy. 2: 259263.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Chlorbufam, 250 mg | sc-227593 | 250 mg | $500.00 |
