CHES (CAS 103-47-9)
QUICK LINKS
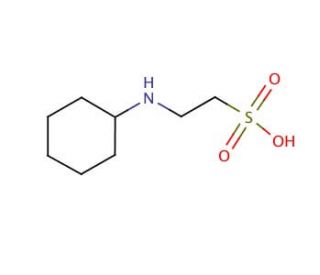
CHES, or 2-(Cyclohexylamino)ethanesulfonic acid, is a zwitterionic organic compound known for its robust buffering capabilities, especially in the alkaline pH range of about 8.6 to 10.0. This makes it particularly useful in biochemical and molecular biology research where maintaining a stable alkaline pH is crucial. CHES is a member of the Good′s buffers, a group of buffers designed for minimal interference with biochemical processes, and it is celebrated for its minimal absorption in the UV-visible spectral range, which is advantageous during spectrophotometric analysis and other light-sensitive procedures. The buffering action of CHES arises from its sulfonic acid group, which can lose a hydrogen ion (proton) and its amine group, which can accept a proton, thereby stabilizing the pH by mitigating changes in hydrogen ion concentration. This dual capacity allows it to effectively maintain environmental conditions in enzymatic reactions where pH stability is necessary for enzyme activity. In research, CHES has been utilized in enzyme kinetics studies and electrophoresis, where precise pH control is needed to ensure the accurate migration of molecules in a gel matrix. Its ability to maintain a stable pH in the alkaline range without interacting significantly with biological molecules or affecting their activity under experimental conditions makes CHES an invaluable tool in scientific studies, particularly those exploring enzymatic mechanisms and properties.
CHES (CAS 103-47-9) References
- Improved detection limit for a direct determination of 8-hydroxy-2'-deoxyguanosine in untreated urine samples by capillary electrophoresis with optical detection. | Tůma, P., et al. 2004. J Chromatogr B Analyt Technol Biomed Life Sci. 813: 255-61. PMID: 15556541
- Suppressed anion chromatography using mixed zwitter-ionic and carbonate eluents. | Chen, Y., et al. 2006. J Chromatogr A. 1118: 3-11. PMID: 16458910
- Analysis of flavonoids by capillary zone electrophoresis with electrokinetic supercharging. | Zhong, H., et al. 2011. Analyst. 136: 4486-91. PMID: 21949941
- Determination of artificial sweeteners by capillary electrophoresis with contactless conductivity detection optimized by hydrodynamic pumping. | Stojkovic, M., et al. 2013. Anal Chim Acta. 787: 254-9. PMID: 23830447
- DFT analysis on the molecular structure, vibrational and electronic spectra of 2-(cyclohexylamino)ethanesulfonic acid. | Renuga Devi, TS., et al. 2015. Spectrochim Acta A Mol Biomol Spectrosc. 137: 761-77. PMID: 25262144
- Crystal structure of the catalytic domain of Clostridium perfringens neuraminidase in complex with a non-carbohydrate-based inhibitor, 2-(cyclohexylamino)ethanesulfonic acid. | Lee, Y., et al. 2017. Biochem Biophys Res Commun. 486: 470-475. PMID: 28315686
- Tuning selectivity in cation-exchange chromatography applied for monoclonal antibody separations, part 1: Alternative mobile phases and fine tuning of the separation. | Farsang, E., et al. 2019. J Pharm Biomed Anal. 168: 138-147. PMID: 30807918
- Ultra-rapid capillary zone electrophoresis method for simultaneous determination of arginine and ibuprofen. | do Prado, AA., et al. 2021. J Sep Sci. 44: 2596-2601. PMID: 33884758
- Affinity labelling of liver alcohol dehydrogenase. Effects of pH and buffers on affinity labelling with iodoacetic acid and (R, S)-2-bromo-3-(5-imidazolyl)propionic acid. | Syvertsen, C. and McKinley-McKee, JS. 1981. Eur J Biochem. 117: 165-70. PMID: 7021155
- Buffers of constant ionic strength for studying pH-dependent processes. | Ellis, KJ. and Morrison, JF. 1982. Methods Enzymol. 87: 405-26. PMID: 7176924
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
CHES, 100 g | sc-216091 | 100 g | $250.00 | |||
CHES, 250 g | sc-216091A | 250 g | $434.00 |
