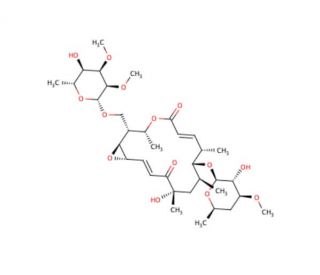
Chalcomycin (CAS 20283-48-1)
QUICK LINKS
Chalcomycin, a polyene macrolide antibiotic, has attracted significant attention in scientific research due to its unique structural features and diverse biological activities. Recent studies have explaind its mechanism of action, revealing its ability to selectively bind to bacterial cell membranes and disrupt their integrity. Chalcomycin exerts its antibacterial effects by forming pores in the lipid bilayer, leading to leakage of cellular contents and ultimately bacterial cell death. This mechanism of action has positioned chalcomycin as a valuable tool for studying membrane biology and bacterial physiology. Moreover, researchers have explored its potential as a scaffold for the design and synthesis of novel antibacterial agents. By modifying the chemical structure of chalcomycin, scientists have generated analogs with enhanced potency and improved pharmacokinetic properties, paving the way for the development of next-generation antibiotics. Additionally, chalcomycin has been utilized as a molecular probe in elucidating the mechanisms of antibiotic resistance in bacteria. By studying the interactions between chalcomycin and resistant bacterial strains, researchers aim to identify novel targets for overcoming antibiotic resistance. Overall, chalcomycin represents a valuable asset in antibacterial research and holds potential for addressing the global challenge of antibiotic resistance.
Chalcomycin (CAS 20283-48-1) References
- Inhibition of the ribosomal peptidyl transferase reaction by the mycarose moiety of the antibiotics carbomycin, spiramycin and tylosin. | Poulsen, SM., et al. 2000. J Mol Biol. 304: 471-81. PMID: 11090288
- Glycosylative inactivation of chalcomycin and tylosin by a clinically isolated Nocardia asteroides strain. | Morisaki, N., et al. 2001. J Antibiot (Tokyo). 54: 157-65. PMID: 11302489
- Production of hybrid 16-membered macrolides by expressing combinations of polyketide synthase genes in engineered Streptomyces fradiae hosts. | Reeves, CD., et al. 2004. Chem Biol. 11: 1465-72. PMID: 15489173
- Structural and functional studies on a 3'-epimerase involved in the biosynthesis of dTDP-6-deoxy-D-allose. | Kubiak, RL., et al. 2012. Biochemistry. 51: 9375-83. PMID: 23116432
- Aldgamycins J-O, 16-Membered Macrolides with a Branched Octose Unit from Streptomycetes sp. and Their Antibacterial Activities. | Wang, CX., et al. 2016. J Nat Prod. 79: 2446-2454. PMID: 27690254
- Exploring the molecular basis for substrate specificity in homologous macrolide biosynthetic cytochromes P450. | DeMars, MD., et al. 2019. J Biol Chem. 294: 15947-15961. PMID: 31488542
- Cross resistance pattern towards anticancer drugs of a human carcinoma multidrug-resistant cell line. | Gupta, RS., et al. 1988. Br J Cancer. 58: 441-7. PMID: 3207599
- Intergenic suppressors of temperature-sensitive sporulation in Bacillus subtilis are allele non-specific. | Sharrock, RA. and Leighton, T. 1981. Mol Gen Genet. 183: 532-7. PMID: 6801427
- The use of PCR to isolate a putative ABC transporter from Saccharopolyspora erythraea. | O'Neill, MP., et al. 1995. FEMS Microbiol Lett. 131: 189-95. PMID: 7557329
- GERI-155, a new macrolide antibiotic related to chalcomycin. | Kim, SD., et al. 1996. J Antibiot (Tokyo). 49: 955-7. PMID: 8931736
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Chalcomycin, 500 µg | sc-391083 | 500 µg | $150.00 | |||
Chalcomycin, 1 mg | sc-391083A | 1 mg | $260.00 |
